Eliminating soluble heavy metal ions from industrial wastewater
The litany of problems and penalties associated with discharging industrial wastewater into sewage treatment systems is a constant concern for today’s industrial manufacturers. The environmental consequences of insufficient or improper wastewater treatment run the gamut from causing corrosion and other interference within those systems to the pass-through of toxic pollutants into surface waters. Add other issues to the mix — including expensive sludge disposal costs and the exposure of workers to toxic substances and hazardous fumes — and it’s easy to understand how devastating the results can be when the importance of wastewater treatment is either deprioritized or ignored.
Neither approach is an option for plants that handle electroplating, electroless nickel plating, printed circuit board manufacturing, metal-forming operations, battery recycling and mining operations. All of these processes (plus others) generate waste streams containing soluble heavy metal ions, and the removal of these ions from wastewater has become a common industrial treatment requirement in modern manufacturing.
Complexors in the alkaline electrolyte chemistry used in the surface-finishing industry make the wastewater extremely difficult to treat through conventional methods.
There are two methods of removing heavy metals from wastewater that are more effective than conventional solutions: precipitation and ultrafiltration. Both can help manufacturers avoid the previously mentioned consequences, but which will deliver the best results? The answer depends on a variety of application-specific criteria that any reputable wastewater treatment system provider will consider before recommending a solution.
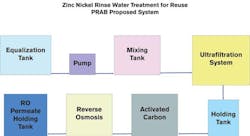
A plating industry example illustrates wastewater treatment variables
In a particular wastewater stream, the chemical form and concentration of soluble heavy metals can vary widely. This is usually dictated by the specific industry that a processing site serves, as well as the mix of operations. In the plating industry, for example, the use of alloy technologies developed by specialty chemical suppliers, such as zinc/nickel and other alloy coatings, has become commonplace to maximize the high corrosion resistance of the metal.
In the plating industry, specialty chemical suppliers have developed alloy technologies and coatings that present wastewater treatment challenges.
Because of the alloy distribution required by the plate thickness throughout the entire part, an alkaline electrolyte chemistry has replaced an acid electrolyte chemistry as the zinc/nickel of choice for today’s surface-finishing industry. Unfortunately, alkaline chemistry contains chelators or complexors (or both) as well as organic additives to allow for the codeposition of the nickel in the deposit with the zinc. The presence of these chelators, complexors and additives makes wastewater treatment extremely challenging using conventional physical or chemical methods.
Precipitation and the use of polymers in a lamella clarifier, followed by a filter press to dewater the sludge, are among the conventional methods that can effectively treat zinc and chrome wastewater. With highly complexed alkaline zinc/nickel, which doesn’t want to precipitate, the chelated metals tend to stay in solution, and the high metal concentrations erase the option of discharging to the local publicly owned treatment works (POTW). Additionally, nickel is a hazardous constituent and is not exempted. Therefore, wastewater from a zinc/nickel process will cause a plant’s filter press cake to become a "listed" RCRA (Resource Conservation and Recovery Act) hazardous waste with the F006 designation, resulting in significant increases to disposal and regulatory costs.
The importance of effectively eliminating solids from wastewater
The solids must be removed from the wastewater as effectively as possible. Typical removal strategies involve precipitating the metals in an insoluble form — such as hydroxides, sulfides, carbonates or some combination — followed by removing the precipitate with conventional clarification or, for very high-quality filtrate, tubular ultrafiltration (UF). The water phase is returned to the ultrafiltration process, while the resultant sludge is collected, thickened and dewatered for landfill disposal. It will still be listed as F006, but at a much smaller volume.
Compared to conventional clarifier techniques, tubular ultrafiltration is a more effective method of ensuring that the treated wastewater stream is free of all of the precipitated metal hydroxide/sulfides/carbonates. The resulting high-quality permeate can be reclaimed in its existing state, fed directly to reverse osmosis equipment for reuse, or discharged to the POTW.
“Compared to conventional clarifier techniques, tubular ultrafiltration is a more effective method of ensuring that the treated wastewater stream is free of all of the precipitated metal hydroxide/sulfides/carbonates.”
It is important to note that while tubular ultrafiltration can process solids concentrations of up to 18 percent (w/w), the most efficient operation normally occurs between 3 percent and 5 percent solids. To produce dewatered solids, typically for landfill disposal, the sludge is normally drawn off and passed through conventional thickening/filter press processes or similar processes. Recycling supernatants from the sludge-handling processes through the tubular membrane filtration (TMF) system helps ensure that the only outputs are the filtered water and the compact solids cake.
Another common method is hydroxide precipitation due to its comparative simplicity. Sulfide precipitation has some advantages, but to minimize the risk of producing toxic levels of hydrogen sulfide gas, pH and oxidation-reduction potential (ORP) must be carefully controlled. Using these two precipitation methods in sequential steps is also an option, particularly where complexes or chelates are present. For some desired outcomes, such as soluble lead reduction, carbonate coprecipitation using sodium or calcium carbonate can also be helpful, as lead carbonate is essentially insoluble (0.00011 g/100 mL at 20°C) and will precipitate out. Phosphate precipitation is another option, although this process is not as common.
Finally, precipitating chelated metals is possible with available membrane technologies that use a proprietary additive in conjunction with a chelate breaker. Among the advantages of this process are:
• Eliminating the need for clarifiers or lamella separators, as well as coagulants for processing due to the small pore size of the microfiltration and ultrafiltration membrane
• A reduction in sludge volume
• Lower labor costs due to a diminished need for constant operator monitoring
• Reduced chemical costs
• A more consistent effluent
Conclusion
When heavy metals are part of a wastewater treatment equation, the whole undertaking can understandably seem intimidating. But it is a challenge that cannot be ignored and must be met head-on, and ideally, in partnership with a proven expert in wastewater solutions for a variety of industries, chemistries and applications. Addressing this important and essential aspect of modern manufacturing not only eases your compliance burden but also brings the added benefit of improved profitability.
Tim Hanna is the vice president of business development for PRAB, a designer and manufacturer of wastewater recycling systems as well as engineered conveyors and equipment for processing turnings, chips, and metalworking fluids. Since 1970, Hanna has worked closely with companies and communities around the world that take advantage of the latest fluid filtration and chip handling technologies to both lower cost and increase environmental stewardship. Visit prab.com.