Granular activated carbons (GAC) are the best available technology (BAT) to purify and protect global drinking water and air supplies. GAC can be manufactured from a wide variety of natural resource materials. Bituminous coal and coconut shells are the dominant raw materials to manufacture GAC for the drinking water application. Demand for GAC is projected to rise 12 percent per year the next five years.1
All activated carbons (AC) are not the same. There are a myriad of different activated carbons and applications. This variable family of activated carbons have fine micro-, meso- and macro-pore structures — a better name is adsorption spaces. They have different adsorption energies (AE), smaller spaces have higher AE and larger adsorption spaces have lower AE. Every adsorbate molecule has a critical AE to remove it from air or water to the GAC adsorptive surface. The micro- and meso-pores are most useful for accumulating water contaminants, 1-5 and 5-50 nanometers respectively. These sized pores can be revealed with the Gravimetric Adsorption Energy Distribution (GAED) full characterization test method.2
Over the last nine years PACS Laboratories has provided many applications for GAED to solve refractory problems.2-19 This paper presents a new application for GAED. GAED is now being used to compare the difference between unused and used GAC in drinking water applications, and other applications, to reveal which pores are filled with adsorbates or chemical impregnants. This GAED information enables better purchasing and used AC change-out decisions. By unused GAC we mean starting GAC, and by used GAC we mean starting GAC which has been through a specific application, such as drinking water or industrial wastewater applications.
What pores are filled?
If you are a potential or actual activated carbon user: Do you know what pores (adsorption spaces) are needed in your application? This is powerful information to get and GAED provides the answer. Most activated carbon users do not know what pores are used in their applications.
Today it is possible for AC users to determine what pores are needed. Knowing what pores are filled in your application provides the opportunity to select an AC with more needed pores. With more useful pores in your applications, expect your finished water to be improved with less AC, which lasts longer. GAED chose GAC, or regular performance monitored GAC, to determine if change-out with fresh GAC provides longer service times.
GAED results executive summary
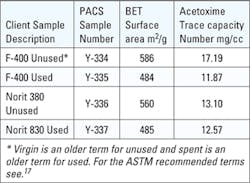
Four client samples of granular activated carbon (GAC) were fully characterized using the Gravimetric Adsorption Energy Distribution (GAED) and ASTM standard test methods. Two samples were unused GAC and two samples were taken from the used GAC drinking water filters.
The unused GAC were Calgon F-400 (Y-334) and Norit 830 (Y-336). The client determined the status of used.
All four samples were compared to each other and a commercially available PCB coconut-based GAC 1250 iodine number. None of the samples had a weight loss greater than 2.3 percent on GAED conditioning, heating the samples in argon at 250° C for 25 minutes, before challenging the sample with 1,1,1,2-tetrafluorethane (TFE). Prior to GAED samples were dried at 150° C for three hours to determine received and dry apparent densities.
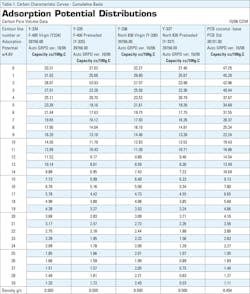
GAED losses of less than eight percent indicate a well-stored sample that has been protected from ambient air moisture pick-up during handling and storage and was also fresh and not oxidized. In terms of total adsorption pore volume, both unused samples outperformed the used version by 16.8 percent and 14.9 percent respectively. See Table 1, the cumulative adsorption space at zero AE in column one.
The difference between unused GAC is only about three percent, and the difference between the used GAC samples is less than one percent. However, the difference in used samples is dependent on materials adsorbed and the degree to which the carbon is saturated.
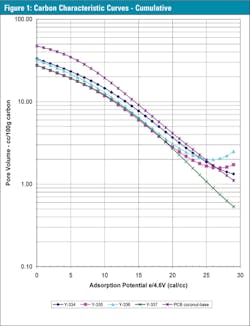
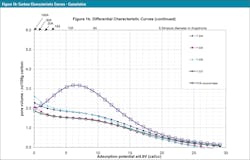
All four samples were similar in terms of the shape of their characteristic curves, see Figure 1. But subtle differences in pore structure at different adsorption energy (AE) levels can be seen in the differential characteristic curves in Figure 1b.
In next month’s issue, we will wrap up the discussion in part two with GAED test method summary, performance prediction models, adsorption isotherms, pore size distributions and conclusions.
About PACS
PACS is celebrating its 29th year of providing laboratory testing and consulting services for the activated carbon industry. PACS announces Call-for-Papers for 2013 International Activated Carbon Conference in Honolulu, Hawaii Feb. 7-8, 2013. Activated carbon courses are provided before and after each carbon conference. All authors can be reached at PACS Testing, Consulting, & Training at 409 Meade Drive, Coraopolis PA. 15108 or phone at 724.457.6576 or www.pacslabs.com.
References- Henry Nowicki, “Demand and Supply for Activated Carbons Next Five Years.” International Activated Carbon Conference. Pittsburgh PA Oct. 4-5, 2012.
- “New Directions in the Activated Carbon Industry.” Henry Nowicki, Mick Greenbank, and Barb Sherman. Filtration News. Vol. 20, No. 5 Sept. 2002. P. 33-39.
- “Advanced Instruments for Sorbent Adsorbate(s) Determination and Sorbent Adsorption and Desorption Evaluation.” H. Nowicki and M. Greenbank. Presented at 23rd Army Science Conference. Orlando FL Dec. 2-5, 2002.
- “New Differential Heat-of-Adsorption Instrument.” H. Nowicki and M. Greenbank. Presented at 23rd Army Science Conference. Orlando FL Dec. 2-5, 2002.
- H.G. Nowicki, et. al. “GAED instrument reveals sorbent materials critical information.” Pittsburgh Conference (PittCon) New analytical Instruments 230-12P, March 2, 2008.
- H.G. Nowicki et. al. “GAED determination of activated carbons in coal fired electric power plants fly ash samples” PittCon Analysis for Energy Production 1570-8P, March 4, 2008.
- H.G. Nowicki, et. al. “GAED location of positional placement of chemical impregnants into Activated Carbons.” PittCon Material Sciences, 2740-5P, March 5, 2008.
- Henry Nowicki, Barbara Sherman, George Nowicki. “GAED Modern Activated Carbon Testing.” Filtration News. August/September 2007 p. 28-33.
- Henry Nowicki, Bud Carr, George Nowicki. “GAED Comparison of Two Activated Carbon Felts” Water Conditioning and Purification. June 2007 p. 28-33.
- Henry Nowicki. “Modern GAED Test Differentiates Activated Carbons”. Water Conditioning and Purification. February 2007 p. 62-64.
- Henry Nowicki and Barbara Sherman. “Activated Carbon: Advanced Test Method.” Filtration News. July/August 2006 p. 14-21.
- Henry Nowicki and Barbara Sherman. “Activated Carbon: Advanced Test Method.” Water Conditioning and Purification. March 2006 p. 32-36.
- Henry Nowicki and Barbara Sherman. “Activated Carbon: Comparison of Classical and Advanced GAED Test Methods.” Water Conditioning and Purification. April 2006 p. 28-35.
- Henry Nowicki. Determination of Adsorption Energy Distributions in Activated Carbons and other Sorbents. 200-20P. Pittsburgh Conference. 2006.
- Henry Nowicki. “Selecting the Best Activated Carbon for the Process Application.” Filtration News. July/August 2005 p. 18-22.
- Henry Nowicki. GAED Helps to Select the Best GAC for a Municipal Drinking Water Plant. 22nd International Activated Carbon Conference. Pittsburgh PA October 7-8, 2008.
- George Nowicki, et. al. “The Importance of Activated Carbon Terminology: A Glossary of Terms” Water Conditioning and Purification. February 2008 p. 88-94.
- Henry Nowicki. “GAED Test provides Advantages over Iodine, Molasses, and BET Surface Area Tests for Purchasing and Used GAC Change-out Decisions.” 30th International Activated Carbon Conference. Pittsburgh PA October 4-5, 2012.
- Henry Nowicki. “GAED and Micromeretics Comparative BET Surface Areas.” 30th International Activated Carbon Conference. Pittsburgh PA October 4-5, 2012.