GAED comparison of unused and used drinking water activated carbons: Part two
Editor’s note: If you missed part one, please go to www.WaterTechOnline.com.
GAED test method summary
Each GAED run measured over 300 adsorption and desorption data points covering seven orders of magnitude in relative pressure (isothermal basis) and three orders of magnitude in GAC loading. The mass adsorbed was also divided by the carbon mass to generate a weight percent loading for easier comparison.
The raw data is plotted in Figure 2. After thermal cleaning the adsorbent gas, C134a or 1,1,1,2-tetrafluoroethane (TFE), is introduced and the loading increases as temperature is decreased and desorption occurs as temperature is increased. Note in Figure 2, the mass loading was plotted against temperature but the relative pressure was also changing. There are three variables affecting physical adsorption performance that change from quasi-equilibrium point to point: Vapor pressure, partial pressure and temperature.
To make comparisons easier, the large data file of adsorption/desorption points at different temperatures and relative pressures was simplified. First the data was interpolated to get 30 evenly spaced points covering the entire TFE adsorption and desorption data acquisition range.
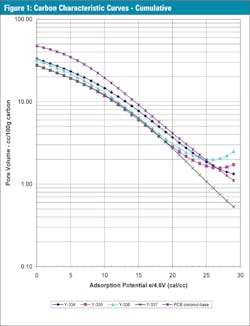
Next the adsorption and desorption results were averaged to get the quasi-equilibrium values (the difference between adsorption and desorption was minimal for these samples — no hysteresis). The y-axis is converted to pore volume measures, in cc liquid adsorbed or cc pores filled per 100 grams sample, instead of weight percent to eliminate adsorbates density differences. The average interpolated volume based data for these characteristic curves is presented in Table 1 and Figures 1 and 1b expanded.
Performance prediction models
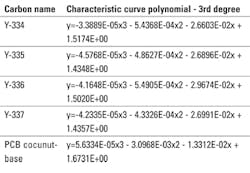
These characteristic curves are the only carbon related information required to predict physical adsorption performance using the Polanyi Adsorption Potential theory. These single and multi-component, gas and liquid phase, computer models are used to predict carbon performance and are available from PACS. To do performance predictions the following polynomials describes these carbon samples:
In the equation, y is the common logarithm of pore volume in cc/100g carbon, and x is the e/4.6V adsorption potential energy in cal/cc.
Adsorption isotherms
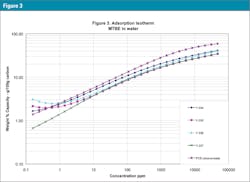
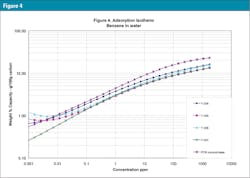
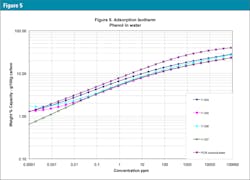
The characteristic curves are also translated into adsorption isotherms using the PACS programs mentioned above: Figure 3 is the isotherm for MTBE (weakly adsorbed material), Figure 4 for benzene (more strongly adsorbed species) and Figure 5 for phenol at pH = 7 (quite strongly adsorbed material). Often isotherms are most important because they tell which GAC is best for their specific applications and how much GAC is needed to clean up the water.
Pore size distributions
The Kelvin equation, modified by Halsey, can be used to convert the characteristic curve data to calculate BET surface areas and pore size distributions. This data is not useful in terms of performance evaluations like isotherms, but some audiences are more comfortable with the concepts of pore radius and a series of capillary sizes when thinking about activated carbon.
Cumulative pore size distribution (Figure 6) were included for this audience. The single and multi-point BET surface area was calculated from these curves.
Conclusion and discussion
The amount of contaminants loaded onto GAC is directly related to the influent concentrations. In drinking water applications the raw water source has low concentrations, which results in lightly loaded used GAC. Used GAC from drinking water plants is relatively easy to furnace reactivate for additional GAC uses. Reactivated GAC costs about half of the original GAC.
GAC is completely used when the influent and effluent are equal. Drinking water GAC applications use 3-20 percent of the original total pore volume in the unused GAC when it is completely used. This total pore volume difference between unused and used GAC is revealed by GAED test method.
In contrast to drinking water applications, industrial aqueous GAC application typically results in use of 20-70 percent of the original total pore volume in the unused GAC. Industrial GAC applications typically have ppm concentrations in the raw water and drinking water has ppb and ppt concentrations in the influent. Raw water concentration differences determine how much of the unused GAC pore volume is filled in the used GAC application.
About PACS
PACS is celebrating its 29th year of providing laboratory testing and consulting services for the activated carbon industry. PACS announces Call-for-Papers for 2013 International Activated Carbon Conference in Honolulu, Hawaii Feb. 7-8, 2013. Activated carbon courses are provided before and after each carbon conference. All authors can be reached at PACS Testing, Consulting, & Training at 409 Meade Drive, Coraopolis, PA. 15108 or phone at (724) 457-6576 or www.pacslabs.com.
Henry Nowicki, Ph.D. has 34 years of broad and deep activated carbon experiences. Henry is a teacher in the PACS Activated Carbon School. Henry’s PACS Short Course is titled “AC Adsorption: Principles, Practices and Opportunities.”
Wayne Schuliger, P.E. technical director for PACS has 48 years of activated carbon experiences. Wayne is a teacher in the Activated Carbon School. Wayne’s PACS Short Course is titled “Design, Operation and Troubleshooting AC Adsorbers.”
George Nowicki, B.S. is an activated carbon chemist at PACS Laboratories for 15 years. George combines business and technical skills for PACS Activated Carbon Services.
Barbara Sherman, B.A. is the manager of operations for 29 years. Barbara directs the day to day PACS Training and Conference business. PACS will host the 31st International Activated Carbon Conference and Courses program in Honolulu, Hawaii Feb. 7-8, 2013.
References
- Henry Nowicki, “Demand and Supply for Activated Carbons Next Five Years.” International Activated Carbon Conference. Pittsburgh PA Oct 4-5, 2012.
- “New Directions in the Activated Carbon Industry.” Henry Nowicki, Mick Greenbank, and Barb Sherman. Filtration News. Vol. 20, No. 5 Sept. 2002. P. 33-39.
- “Advanced Instruments for Sorbent Adsorbate(s) Determination and Sorbent Adsorption and Desorption Evaluation.” H. Nowicki and M. Greenbank. Presented at 23rd Army Science Conference. Orlando FL Dec. 2-5, 2002.
- “New Differential Heat-of-Adsorption Instrument.” H. Nowicki and M. Greenbank. Presented at 23rd Army Science Conference. Orlando FL Dec. 2-5, 2002.
- H.G. Nowicki, et. al. “GAED instrument reveals sorbent materials critical information.” Pittsburgh Conference (PittCon) New analytical Instruments 230-12P, March 2, 2008.
- H.G. Nowicki et. al. “GAED determination of activated carbons in coal fired electric power plants fly ash samples” PittCon Analysis for Energy Production 1570-8P, March 4, 2008.
- H.G. Nowicki, et.al. “GAED location of positional placement of chemical impregnants into Activated Carbons.” PittCon Material Sciences, 2740-5P, March 5, 2008.
- Henry Nowicki, Barbara Sherman, George Nowicki. “GAED Modern Activated Carbon Testing.” Filtration News. August/September 2007 p. 28-33.
- Henry Nowicki, Bud Carr, George Nowicki. “GAED Comparison of Two Activated Carbon Felts” Water Conditioning and Purification. June 2007 p. 28-33.
- Henry Nowicki. “Modern GAED Test Differentiates Activated Carbons”. Water Conditioning and Purification. February 2007 p. 62-64.
- Henry Nowicki and Barbara Sherman. “Activated Carbon: Advanced Test Method.” Filtration News. July/August 2006 p. 14-21.
- Henry Nowicki and Barbara Sherman. “Activated Carbon: Advanced Test Method.” Water Conditioning and Purification. March 2006 p. 32-36. 13. Henry Nowicki and Barbara Sherman. “Activated Carbon: Comparison of Classical and Advanced GAED Test Methods.” Water Conditioning and Purification. April 2006 p. 28-35.
- Henry Nowicki. Determination of Adsorption Energy Distributions in Activated Carbons and other Sorbents. 200-20P. Pittsburgh Conference. 2006.
- Henry Nowicki. “Selecting the Best Activated Carbon for the Process Application.” Filtration News. July/August 2005 p. 18-22.
- Henry Nowicki. GAED Helps to Select the Best GAC for a Municipal Drinking Water Plant. 22nd International Activated Carbon Conference. Pittsburgh PA October 7-8, 2008.
- George Nowicki, et. al. “The Importance of Activated Carbon Terminology: A Glossary of Terms” Water Conditioning and Purification. February 2008 p. 88-94.
- Henry Nowicki. “GAED Test provides Advantages over Iodine, Molasses, and BET Surface Area Tests for Purchasing and Used GAC Change-out Decisions.” 30th International Activated Carbon Conference. Pittsburgh PA October 4-5, 2012.
- Henry Nowicki. “GAED and Micromeretics Comparative BET Surface Areas.” 30th International Activated Carbon Conference. Pittsburgh PA October 4-5, 2012.