Monitoring Common Ions During Desalination Using Ion Chromatography
BY LIPIKA BASUMALLICK, DEANNA HURUM AND JEFF ROHRER
Desalination technologies are an attractive alternative to produce water from salty or brackish water to meet drinking and irrigation needs. As of 2009, there were 14,450 desalination plants worldwide, producing more than 60 million cubic meters of water a day.
A wide variety of techniques are currently available and also under development for desalination. Most of these use distillation or membrane techniques. The performance of desalination processes is evaluated by monitoring the common anions and cations in the feed, intermediate, and final water product.
For drinking water, ion chromatography (IC) is approved for monitoring primary and secondary anions according to U.S. Environmental Protection Agency (EPA) Method 300.02, and U.S. federal and state regulatory agencies ensure that the U.S. National Primary and Secondary Drinking Water Standards are met.
Common cations, though not considered as contaminants, are monitored and reported by many public water suppliers in the U.S., European countries, and Japan. Cations, particularly calcium and magnesium, are measured to determine water hardness. During desalination, the levels of divalent cations affect the performance of membrane processes like reverse osmosis. High levels of calcium or magnesium result in frequent membrane fouling, which is highly undesirable. It is thus critical to monitor anions and cations at all stages of desalination.
New Testing Method
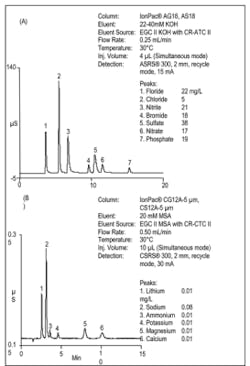
An IC method has been developed that uses anion-exchange and cation-exchange columns and suppressed conductivity detection to simultaneously measure the common anions and cations in water samples obtained from desalination processes. This proposed method uses a 2 mm column format for anion separations, a 3 mm column format for cation separations, and electrolytically generated eluents. It requires only the addition of deionized water for continuous operation. The smaller column diameters reduce eluent use and waste generation, and thus operating costs. This IC method supports all the monitoring needs of a desalination facility as it can measure anions and cations in diverse matrices ranging from seawater to drinking water.
Table 1. Linear Range, Precisions, Recoveries for Anions and CationsExperimental
During testing, all data were collected on a Dionex ICS-3000 system that consisted of dual pumps, an eluent generator, and a detector/chromatography module. The system was configured for simultaneous injection into the anion and cation detection systems. One control panel monitored and controlled both systems and all samples in one sequence.
Using a Dionex Reagent-Free™ IC (RFIC™) system, the common anions and cations are easily resolved and measured in 20 minutes (Figure 1A and 1B). Note that this method provided good resolution between sodium and ammonium, the two analytes that are challenging to resolve, especially when one is in a large excess relative to the other. This method also achieved good retention of fluoride, which was well resolved from the void volume so that its measurement would not be compromised from compounds that are not retained by the anion-exchange column.
The linearity of the method for the common anions and cations was determined by triplicate injections of calibration standards in deionized water. The concentration range for each ion was chosen to cover the average concentration range of the ion in drinking water and diluted seawater (Table 1).
Correlation coefficient values obtained from the calibration plots were between 0.9994 and 0.9999, suggesting that this method enables the quantitative analysis of a wide range of waters, from diluted seawater to drinking water. It is interesting to note that the calibration curves are linear for all anions and cations except ammonium. Analytes that form weak acids or bases in the suppressor are known to exhibit non-linear behavior. A quadratic curve fitting function was better for ammonium.
High retention time precisions (<0.01% - 0.07%), and high peak area precisions (0.04% - 0.97%) were achieved with this method. This is attributed to the consistent generation of high purity eluents using the eluent generator modules and the continuously regenerated trap columns.
This method was used to analyze a water sample from the San Francisco bay in California, which is representative of a typical feed into a desalination plant. The bay water sample was diluted 200 fold so that measured concentrations were in the calibrated ranges. The major inorganic anions in bay water are chloride and sulfate, and the major inorganic cations are sodium, potassium, magnesium, and calcium. All anions and cations are well resolved and have acceptable recoveries (80-120%, Table 1) using the criteria outlined in U. S. EPA Method 300.0.
Using the current method, tap water samples showed fluoride, chloride, and sulfate as the predominant anions, and sodium, calcium, and magnesium as the major cations. Again, all anions and cations are well resolved and, with the exception of nitrite have acceptable recoveries (80-120%, Table 1). The low recovery of nitrite could be attributed to biological activity in these samples (which is minimal in the high saline matrices) and the instability of nitrite in oxidizing environments, such as chlorinated water or other oxidizing disinfectants in drinking water.
Conclusion
In summary, the robust water monitoring method provided good resolution, high sample throughput and acceptable recoveries for common anions and cations in diverse matrices ranging from seawater to drinking water. The setup requires only water for continuous operation, generates less waste and uses less eluent, saving both time and money.
About the Authors: Lipika Basumallick, Ph. D., is a Staff Chemist at Dionex Corp., Sunnyvale, CA. Deanna Hurum, Ph.D., is a Staff Chemist and Jeff Rohrer, Ph. D., is the Director of Corporate Applications Development at Dionex in Sunnyvale.