Membrane Contactors Improve Performance of Conventional Ion Exchange, EDI Systems
by Fred Wiesler and Beth Kitteringham
For decades ion exchange beds have been used to produce deionized water. In a simplistic summary, the ion exchange bed will preferentially exchange a H+ or OH- ions for other ions in the water. Over time the ion exchange resin becomes saturated with the ions in the water and it can no longer perform the exchange. Once this occurs, the ion exchange resin is regenerated. Cation resin is regenerated with acid and anion resin is regenerated with a base.
Carbon dioxide ionizes in water and adds a load onto the anion exchange resin bed. Sodium hydroxide is typically the most expensive chemical used in the regeneration process. By removing the carbon dioxide, the volume of resin and amount of sodium hydroxide used for the regeneration can be reduced. This results in a lower capital and operating cost.
Carbon dioxide can be removed from the water in a deaeration process. As a general rule of thumb deaeration technologies can be economically justified when the water flow rate exceeds 50 gpm and the inlet carbon dioxide is greater than 50 ppm. Under these conditions, the added capital and operating costs of the tower begin to offset the chemical consumption and disposal of the regeneration chemicals.
In some demanding applications such as water systems in pharmaceutical plants, deaeration towers are not recommended due to the exposure of the water to air that may contain biological contaminants.
Membrane Contactor systems are suitable for use in applications where deaeration towers are not used due to possible air contamination. The membrane used in a membrane contactor acts as a barrier between the air and water stream and any contaminants are not able to reach the liquid stream. The capital and operating costs of a membrane contactor system are also lower than a deaeration tower so the economics can be easily justified.
Conventional ion exchange systems are slowly being replaced by EDI and CEDI technologies. CO2 loading is a problem for both technologies. Membrane contactors offer a lower cost and contamination free process to remove the carbon dioxide from the water. The emphasis in the following paragraphs will be on CO2 removal in conjunction with EDI technology.
Electrodeionization
Electrodeionization (EDI) is a widely used water treatment process. The EDI technology is an electrochemical process that uses ion selective membranes and an electrical potential to continuously remove ions from water. The process uses ion exchange resin to remove the ions from the feed stream, producing pure water. A DC current continuously regenerates the resin.
This technology operates under the same principles as conventional ion exchange resin technology with the added benefit of being free of regeneration chemicals. This eliminates the need to take the resin off line for regeneration. This can provide a significant reduction in operating costs and is a positive environmental protection step.
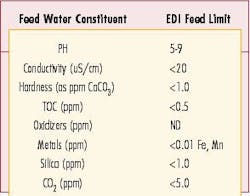
In order to maximize the performance of an EDI unit, proper pretreatment is required. Suppliers of EDI equipment have different guidelines regarding feed water requirements but generally the specification can be summarized as follows:
Carbon Dioxide and Conductivity
When ion exchange is used to polish reverse osmosis membrane (RO) permeate, carbon dioxide needs to be controlled. As mentioned, this is true for both conventional mixed bed ion exchange and EDI technologies. Excessive carbon dioxide is the leading cause of an EDI system not meeting design specifications. Carbon dioxide gas dissociates in water to form HCO3- and CO32-. These ionic species will contribute to the total anionic load and should be added to the measured total. Overloading the anionic capacity of the EDI unit will lead to higher product conductivity and higher levels of weakly charged ions like boron and silica in the product water.
Feed water conductivity does not show a complete picture of the total ionic load in a water system. Conductivity measurement devices do not detect the full amount of weakly ionized species like carbon dioxide and silica. Suppliers have developed methods to quantify the total ionic load on the EDI system. Two methods are described below:
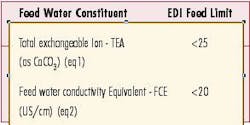
TEA as ppm CaCO3 (Total exchangeable Ion) = TDS (ppm as CaCO3, based on ionized species minus HCO3-)+(ppm CO2*1.14*1.7) +(ppm HCO3- as CaCO3*1.7)
FCE as US/cm (Feed water Conductivity Equivalent) = (conductivity + ppm CO2*2.66+ppm SiO2*1.94)
These calculations show that the inlet carbon dioxide will contribute a significant anionic load on the EDI system. For example if the inlet water contains 5 ppm CO2 and 1.5 ppm bicarbonate (HCO3-) as CaCO3 this will add 12.24 ppm as CaCO3 to the TEA and 13.3 Us/cm to the FCE. The most economical way to lower the load on the EDI is to remove a portion of the carbon dioxide.
EDI Pretreatment
EDI feed-water pre-treatment is typically made up of softening and filtration using a reverse osmosis membrane system. These technologies can effectively remove particles, TOC, ions, and dissolved minerals from the water. Dissolved gases, however, are not effectively removed with these technologies.
Dissolved gasses such as carbon dioxide can be removed using forced draft deaeration towers. Such towers are effective but can add unwanted particulate and biological contamination into the water. These systems also require a bigger footprint and repressurization pumps to get the water to the next step in the process.
Carbon dioxide can also be removed by reverse osmosis by adjusting the pH of the feed water to the RO. Carbon dioxide gas ionizes under high pH conditions and can be partially rejected by the reverse osmosis membrane. This technique is an effective way to remove carbon dioxide but requires chemical addition to shift the pH. It also requires softening before the RO to prevent scaling at the higher pH. In addition, the carbon dioxide level in the water will change based on seasonal variations and may require special controls to properly control the chemical addition.
Membrane Contactors
Liqui-Cel® Membrane contactors, offered by Membrana, have been used to remove dissolved gas from water for over 10 years. They are used in Power, Semiconductor, Pharmaceutical, food & beverage and industrial applications to remove dissolved oxygen and carbon dioxide from water. New lower cost high-capacity industrial grade devices have been developed to make removing dissolved carbon dioxide from water with modular membrane devices even more affordable.
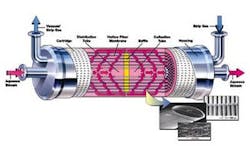
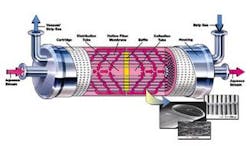
The Liqui-Cel Extra-Flow Membrane Contactor design contains thousands of microporous hollow fibers. These fibers are knitted into a fabric that is wound around a distribution tube with a central baffle.
During operation, the liquid flows over the shellside (outside) of the hollow fibers. The patented Extra-Flow design incorporates a baffle in the middle of the contactor, which directs the liquid radially across the membrane fibers to maximize membrane surface area. A strip gas or vacuum, either separately or in combination, is applied on the lumenside (inside) of the hollow fibers and this provides the driving force to move the gas from the liquid phase into the gas phase.
Because of its hydrophobic nature, the membrane acts as an inert support to allow direct contact between a gas and liquid phase without dispersion.
By adjusting the pressure and concentration of the gas in contact with the liquid, dissolved gasses can be removed from water. A typical membrane contactor system designed for carbon dioxide removal flows water on the outside of the membrane while room air is drawn into the device under a vacuum on the other side. The gasses freely pass from the liquid phase through the membrane pores and are carried away by the vacuum or strip gas phase.
Membrane contactors are available in a wide range of sizes. They are designed for small flow applications of 0.1 gpm to over 4000 gpm. They operate in line under pressure and do not require surge tanks or transfer pumps. They can be installed downstream of the RO and upstream of the EDI unit to lower the carbon dioxide level to < 1 ppm.
Membrane contactors can be designed to lower dissolved carbon dioxide to virtually any level required by system needs. Practically, the carbon dioxide inlet is typically reduced to 1-5 ppm. A secondary benefit of lowering the anionic load is a power savings. As the ionic load is decreased, the power consumption of the EDI system can be reduced. This can lower the operating cost of the EDI system. A third benefit of lowering the anionic load is improving the removal of the weakly charged anions like silica and boron.
About the Authors: Fred Wiesler is a technical sales manager in the Industrial Separations Group of Membrana, located in Charlotte, NC, who is currently responsible for sales and service of Membrana's Membrane Contactor product line for Asia. Wiesler has more than 15 years experience in the field of membrane separations and holds patents on membrane contactor designs. Beth Kitteringham is the marketing manager for the Industrial Separations Group of Membrana. She has more than 10 years of experience in water treatment in various technical sales and marketing positions and she is also located in Charlotte.