Physicochemical Treatment of Water Helps Control Scaling
By David McLachlan
Physicochemical water conditioning methods — that do not use chemical additives — can be used to control scaling formations experienced in re-circulating water systems.
Scale is usually formed by precipitation of calcium and magnesium carbonate, which becomes increasingly less soluble at higher temperatures. Scale formation resulting from the dissolved minerals in water can be expensive and its treatment can have long lasting and dire consequences to a system as well as to the environment.
The operational impact of deposited scale can result in reduced fluid flow, loss of heat transfer capability, increased system corrosion, and enhanced bio fouling. Chemicals used to control scale formation are hazardous, polluting, and costly and in many cases can be eliminated.
Minerals dissolved in water that results in scale can be controlled in two ways. First by the removal of the ions before water is used, or secondly by leaving the dissolved minerals in the water and treating the water in order to prevent or control scale formation.
The removal of both suspended and/or dissolved solids from waters can be accomplished by water softening, flocculation, reverse osmosis, etc. These techniques may not be economical at the point of use in industrial systems. In large industrial water re-circulating heat exchange systems, the emphasis should be placed not on the removal of ions or suspended solids, but on leaving the dissolved solids (ions) in the system and using techniques that inhibit suspended and dissolved solids from forming scale.
For the reader who wants to have a deeper understanding of the physicochemical mechanisms, there are three additional components that are useful, but are outside the scope of this article. They are; 1) that all chemical reactions are controlled by the outer electrons of reactant atoms, 2) that crystal growth (scale formation) is primarily a single atom process, and 3) that colloidal particles (small crystalline carbonates) normally carry a negative surface charge and are quite stable.
The following four physicochemical water treatment techniques were selected for discussion because they do not add or subtract chemicals from the water being treated.
Catalytic Processing
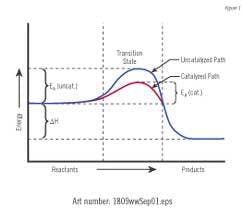
A catalyst is a compound which increases the rate of a chemical reaction but which is not consumed by the reaction. Catalyst provides alternate paths by which a higher proportion of the reactants (ions) will have sufficient energy to form products (scale). The kinetics or rate of the reaction can be greatly increased by the presence of the catalyst.
Honeywell's kaltecpro™ uses a catalysis principle that generates a large number of stable seed crystals (colloid particles). Friction forces from liquid flow then shear off the newly formed crystals from the kaltecpro™ beads. These small crystals are of sufficient size to be stable and serve as 'seed crystals' for further precipitation. The products derived from this technology are primarily directed toward the residential and commercial markets for potable hot water systems.
Magnetic
History shows that Egyptians used magnets two to three thousand years ago in an attempt to reduce scale deposits. Lodestones were used in the 19th century in cooking and laundry applications. The use of magnets may be one of the oldest methods in the prevention of scaling.
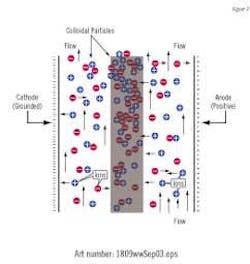
Hard water contains ions, both positive and negatively charged. When a moving ionized fluid passes through a magnetic field, a force is exerted on each ion (Lorentz Force). This force is perpendicular to both the magnetic field and the direction of motion and is proportional to the velocity of the ions. Since the force is at right angles to the velocity, it will not affect the magnitude of the velocity nor its kinetic energy but will merely alter its direction. The forces on ions of opposite charges are in opposite directions and tend to increase the frequency with which ions of opposite charge (Ca++ and CO3—) come into contact with each other and form mineral precipitates, (colloidal particles).
The marketing of magnetic devices is generally directed at smaller flow applications (residential). However, Fluidyne International Inc., services boilers between 10 and 600 hp and flows up to 200 gpm with permanent magnets with good success.
Induced Electric Field
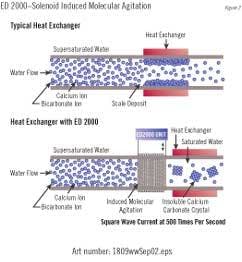
"Solenoid-Induced-Molecular Agita-tion" uses a time varying electronic current in a solenoid wrapped around a pipe to create an induced electromagnetic field inside the pipe. Dr. Young Cho of Drexel University developed this technology (marketed by York International, Applied Water Technologies, Inc., etc.). It has the capability of treating large industrial flows.
When charged ions such as calcium and carbonate ions pass through the solenoid, they experience an induced pulsating electrical field (Faraday's law of induction). The induced electric field provides the necessary molecular opposing redirection of the charged mineral ions, increasing the number of collisions and hence nucleation of small precipitate (colloidal particles).
Electrostatic
Electrostatic water treatment technology got its start in 1957, when Roy McMahon began his experimentation and development of electrostatic devices for fluid treatment. The principal suppliers of this technology are Electrostatic Technologies, Inc. (FluidTron® Systems, a direct descendent of McMahon's Company), Zeta Corporation (Zeta Rod®) and York Energy Conservation (Ion Stick®).
Electrostatic water treatment technology is based on a strong electric field between a charged electrode and a ground electrode similar to that found in a capacitor. The strong and highly focused applied electric field (up to 30KV-dc) causes the movement of charged ions. This movement results in localized concentration gradients and the redirection of the velocity of the ions, increasing their respective kinetic energy and the frequency of collision between cations (Ca++) and anions (CO3—). These factors create a condition favorable for nucleation and the formation of stable colloidal particles.
If colloidal particles (scale) are formed in the cooler portion of the re-circulating system, the decreased solubility in the warmer section will give the necessary driving force for possible additional precipitation. Due to the much larger surface area of the now existing colloidal particles compared to the walls of the heat exchanger the scale growth on these surfaces will be minimized. Conversely, if the degree of saturation is low, descaling can take place. In a cooling tower environment, this enhanced nucleation and subsequent growth control allows for a slightly elevated pH, normally between 8.2 and 9.5, leading to an improved corrosion and bio-fouling environment.
Electrostatic treatment systems are capable of handling very high fluid flows. This allows the application of these products across a wide spectrum, including commercial and heavy industrial installations.
Summation
Chemical reactions are controlled by the electrical charges contained within atoms and these electrical characteristics affect the behavior of water and minerals in solution or suspension. There is an intimate relationship between magnetic and electrical fields. An electrical field, induced or applied directly, can create stable changes in a water/mineral solutions through the formation of small insoluble calcite crystals at moderate temperatures. The particles are powdery and fluffy, easily removed by turbulence and routine blow down. The level of saturation of the water significantly decreases; thus, new scale deposits on the heat transfer surface are prevented.
Since water chemistry is complicated and in special cases difficult to control, empirical data must be obtained to ensure that the correct water treatment regiment is in position for each installation. At the onset of a different water treatment system, frequent monitoring and regular follow-up is required to insure optimum control of scale, corrosion, and bio-fouling.
Physicochemical systems correctly applied in large flow industrial setting can have a very positive impact. There are several advantages to the use of these systems, including operational savings, the elimination or reduction of hazardous chemicals, and water conservation resulting in an environmentally friendly water treatment system.
About the Author: David McLachlan is vice president of Fluid Treatment Solutions, Inc., with 30 years of industrial experience. He holds a Ph.D. In Metallurgical Engineering specializing in process development. Ph. 913-342-0110, email - [email protected]