Production of HPW/WFI pusing RO, EDI and Ultrafiltration
• Integrity of process water system in the pharmaceutical water plant is assured by packaged, skid-mounted high purity system for ultra-pure water
by Reune Runyon
Although most companies use distillation to produce water-for-injection (WFI) grade water the use of membrane processes in the production of WFI quality, highly purified water (HPW), for the pharmaceutical industry allows cost reductions by a factor of 5.5 to 7.5 in comparison to distillation depending on the quantity of water required. The use of reverse osmosis (RO) in combination with electro-deionisation (EDI) and a subsequent RO or ultrafiltration (UF) step is thus a clear financial benefit in controlling costs. The chemical and physical characteristics of water produced in this way comply with the U.S. Pharmacopoeia (USP) requirements for WFI. In Europe, this production method may be used to generate HPW in compliance with the European Pharmacopeia (EP) specification.
With Osmotron Inject, ultra-pure water specialist Christ, a member of the Christ Water Technology Group, introduces a plant for the production of HPW by means of two RO stages and EDI. The compact unit is entirely hot water-sanitisable and constitutes a safe, closed system. For applications calling for an integrity test, a plant configuration replacing the second RO stage with a UF stage is preferably used.
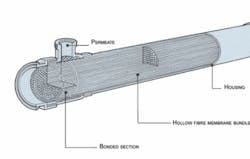
In both configurations, after having passed the security filter, softened water reaches the RO stage at a high pressure as the first process step. The major part of the water passes through the membrane with a residual salt content of approximately 2%, leaving most of the dissolved and any suspended matter behind. In the next step, the permeate passes to the patented, temperature-stable Christ Septrosan EDI module, where it’s fully demineralised. This treatment also reduces pyrogens and microbial contamination to a great extent due to the very high pH gradients of pH 2 to 12. The last process step is RO or UF. When an integrity test is required a UF stage is used. The hollow fibre UF module is also sanitisable and can be completely drained. The molecular weight cut-off (MWCO) rating used is 6,000 Dalton. The double-skinned asymmetrical membranes and the deadspace-free, entirely resin potted, construction offers the highest security. To integrity test the UF modules the concentrate (reject water) chambers are emptied and pressurised air is applied. A deficient module is identified when air bubbles are observed moving upwards on the filtrate side. The UF process is also advantageous since it allows a yield of 100%.
As with the Osmotron Inject version using a terminal RO step, the Osmotron Inject UF version also produces a water quality considerably better than the limit values of USP 27 and EP 5. The conductivity reaches a value of approximately 0.1 µS/cm and the TOC value is typically less than 20 ppb. The relevant microbial values are typically less than 5 CFU/100 ml and endo¬toxins levels less than 0.06 EU/ml. The standard product range provides 500 to 10,000 litres per hour and customised configurations are available for higher flowrates. The systems are manufactured using 316 L, orbitally welded sanitary pipework with a surface roughness Ra of less than 0.8 µm.
About the Author: Reune Runyon is a global key account manager for Christ AG, based in Aesch, Switzerland. Contact: +41-61-755 83 64 or [email protected]