Protecting Water and Wastewater Infrastructure from Corrosion
by Philip G. Rahrig
Whether over water, submerged in water or exposed to moisture-laden air because of close proximity to fresh or salt water bodies, steel is acutely susceptible to corrosion. With a variety of corrosion protection methods from barrier to cathodic based on long-term performance and economy, hot-dip galvanizing is regularly selected. This article dissects the performance of hot-dip galvanized steel in freshwater, salt water, warm water, cold water, tidal zones and above-water applications.
Atmospheric Conditions
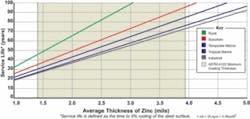
Galvanized steel performs best when the zinc coating is permitted to naturally react with oxygen, hydrogen and carbon dioxide in the air to form a stable zinc carbonate patina. In atmospheric exposure, this zinc carbonate film is inert, not water soluble, and is generally slowly removed over long periods of time only by the physical forces of abrasion and erosion (wind, water) or chemical reactions. In the case of atmospheric corrosion, thousands of galvanized structures have been tested over many decades and the life expectancy of the zinc coating without any maintenance can be accurately predicted (see Figure 1). As the chart indicates, corrosion prevention without any maintenance for 35-40 years in marine atmospheres is easily achievable by galvanizing. Factors affecting performance include sulfides and chlorides in the air.
Above-Water Applications
Bridging the gap - The Stearns Bayou Bridge in Ottowa County, MI, was constructed in 1966. This fully galvanized 420-foot long bridge, is just six feet above freshwater and subject to winter de-icing salts. Upon recent inspection, the principal steel had a projected life expectancy until first maintenance of 66 years.
Clarifying matters - In 1989, the Pittsburgh Water Treatment Plant used 250 tons of hot-dip galvanized steel to construct the truss network covering the four-basin clarifier. Harsh chemicals added to the clarifier in the flocculation process (chlorine, alum, lime) accelerate the corrosion process on unprotected steel. A 1997 inspection of the zinc coating of the galvanized steel measured 5.0 mils of zinc, enough to deliver at least 50 more years of maintenance-free performance.
Avoiding Murphy’s law - On the western edge of Lake Superior in Duluth, MN, the Murphy Oil Terminal was designed with hot-dip galvanized steel to eliminate costly maintenance that disrupted operations and allow for a quick hose-down of all areas in the event of spills. The stairs, railings, platforms, and structural steel in the dockside crane, tank farm and interior equipment mezzanine areas were galvanized.
Zinc Corrosion Rates
In the case of chemical reactions in water, predictability of the effectiveness of corrosion prevention of a galvanized system is much more difficult because of myriad variables associated with exposure. Among those variables are pH level, oxygen content, water temperature, water condition (hard and soft), water type (de-ionized, fresh, salt), agitation and tidal exposure.
De-ionized (pure) water has an aggressive corrosive effect on hot-dip galvanized zinc coatings and even more so when it’s aerated. As oxygen content increases, so too does the corrosion rate of zinc. In fact, oxygen is five to 10 times more corrosive to zinc than carbonic acid.
Freshwater environments have two major constituents for categorizing zinc corrosion potential: hard and soft water. Carbonates and bicarbonates, present in some concentration in freshwater, tend to deposit protective films on the zinc surface, stifling corrosion. Carbonates subdue the corrosion effects of anions, the most corrosive to zinc being chloride in concentration of 50 mg/L or more. The softer the water, the lower it is in carbonate and thus a more pronounced chloride content and higher corrosion rate. Conversely, the harder the water, the greater the carbonate level and corrosiveness of chlorides is minimized. Therefore, the general rule is that soft water is more aggressive to hot-dip galvanized steel while hard water is passive.
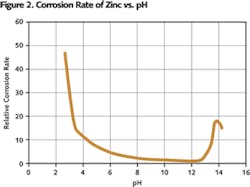
Seawater is high in salt content in the form of sodium chloride. Typical surface seawater has a pH of 8, due to excess amounts of carbonates. The pH may fall to 7 in stagnant waters. Depth of water also plays a role in pH determination. The pH decreases with depth. Since zinc is amphoteric, it is soluble in both base and acidic solutions. However in the broad range of pH from five to 13, zinc is most stable and has a very low corrosion rate (see Figure 2).
Temperature also affects zinc’s corrosion rate. Seawater temperatures can vary widely from 28.4°F (-2°C) at the earth’s poles to 95°F (35°C) near the equator. The higher the temperature the greater dissolution of zinc in water. Tropical seawater yields higher corrosion rates, especially in polluted waters.
Tidal zones and fluid agitation are also important considerations in determining corrosion protection delivered by hot-dip galvanized steel. Often, this motion of “washing” the carbonates off the zinc surface and not allowing them to form a protective film, along with zinc erosion, is the cause for base steel corrosion.
Submerged Applications
Dammed if you don’t - The U.S. Army Corps of Engineers used 105 tons of hot-dip galvanized steel trash racks and fish screen frames in 1997 on six dams along the Columbia and Snake rivers in Oregon and Washington to dramatically reduce the kill rate of salmon and steelhead smolt swimming through the dams from 77% to 12%. Catwalks and dam-top grating, dam locks, handrails, fish diversion outlets and fish ladders all showed no signs of wear or corrosion during a recent inspection.
An industrial approach - Over 40 years of near maintenance-free service is the experience of the Back River Waste Treatment Plant, designed to treat corrosive sludge and industrial wastes of the Baltimore area. Galvanized weirs in settling tanks have seen continuous service since the plant opened in 1938. In the final settling tanks, weirs, brackets and guide rails have remained in excellent condition for 30 years. In the trickling filter system, galvanized steel rotary distributors have been maintenance free for over 30 years as well. And the galvanized grit-removal gratings served 40 years before they required removal, re-galvanizing and re-installation.
Avoiding invasive species - A recent study in the Great Lakes area showed that hot-dip galvanized steel experienced a much lower rate of zebra mussel fouling. Test panels deployed at Oak Orchard Creek in Medina, NY, during 1997 and 1998 by the Industry/University Cooperative Center for Biosurfaces demonstrated that hot-dip galvanized steel would develop a coating of zebra mussels at a much reduced rate from concrete, bare steel and uncoated PVC.
Conclusion
The performance of hot-dip galvanized steel in above-water and close-proximity conditions is well documented and predictable. Maintenance-free service life of 50 years or more is common. Determining the corrosion rate (life expectancy) of galvanized steel’s zinc coating in water isn’t as easily determined or predictable. Soft water is a harsher environment to zinc than is hard water or even cold climate seawater. Temperate seawater deposits protective scales on zinc and is less corrosive than soft water. High oxygen content and warm waters can be detrimental to zinc as well. Corrosion of zinc is lowest in the 5-12 pH range. And most natural potable waters have a pH range of 5-8.5. The corrosion of zinc in such waters, though, is accelerated largely by impurities present in the water, and rarely is natural water pure.
Anything that disturbs formation of a protective film (zinc carbonate) on the zinc surface will inhibit the hot-dip galvanized coating from delivering superior corrosion protection. Other factors - including pH, time of exposure, temperature and fluid agitation - also influence aqueous corrosion of zinc in water. Having identified variables that adversely affect the galvanized coating, it’s important to note galvanized coatings on steel used in submersed applications are still one of the best methods of corrosion prevention. It’s common for hot-dip galvanized steel to perform flawlessly in seawater for 10 years or more.
About the Author: Philip G. Rahrig has been executive director of the Centennial, CO-based American Galvanizers Association (www.galvanizeit.org) for over eight years. The AGA delivers zinc, corrosion theory and galvanizing information to the architectural and engineering communities. Rahrig has a degree in business from Cincinnati’s Xavier University and physics from Thomas More College, in Ft. Mitchell, KY, as well as 10 years of experience in the steel industry with USX.