Boiler Feedwater & HRSG: A New Method for Exposing Condenser Leaks – On-Line Particle Detection
by Robert L. Bryant
Anyone involved with steam/water operations in power plants can appreciate the significance of a cooling water leak into returned condensate. Leaks can occur under normal operation, and can certainly be a result of corrosion due to poor water chemistry. The severity of problems caused by such contamination escalates with higher boiler operating pressures, primarily due to the very sensitive chemistry “balance” needed to prevent tube deposits, corrosion, and/or metal cracking. Even very small amounts, say at the parts per billion (ppb) level, of certain non-water substances can cause upsets in the boiler water chemistry resulting in severe problems, short and long term. Although condensate polishers can remove many contaminants entering the system when tube leaks occur (cations, anions, organics, etc.), the logical approach should be:
1. Continuously monitor individual condensate streams graphically, and “trend” results to determine if a leak occurs
2. Isolate area where leak is occurring, if possible
3. Define relative size of leak, and the rate at which it’s increasing/decreasing with time, varying loads, etc.
Historically, cation conductivity has been the standard method for condenser leak detection. Specific ion analysis is another approach. This paper presents an alternate, or perhaps complementary, method for condensate monitoring consisting of particulate measurement.
Measuring Instruments
A. Conductivity – Conductivity is the measurement of a liquid’s ability to transfer electrical current. In power plants, conductivity instruments are used to measure contaminants (consisting primarily of mineral salts) in high-purity water. Air intrusion into surface condensers, and the injection of other treatment chemicals such as ammonia, amines, etc., however, can affect conductivity. These instruments cannot differentiate between minerals and gases.
There are two types of conductivity measurements: specific and cation. Specific conductivity can detect only large amounts of contaminants. Condenser tube leaks normally start out very small, and may increase over time. For example, condensate flow may be 5,000 gpm, and the “leak” 0.1 gpm. On a volumetric basis, this is 20 ppm. Specific conductance instruments may not be able to detect this small leak unless the cooling water conductivity is very high to start.
Cation conductivity instruments pass the condensate sample through a cation (ion exchange) resin to convert cations (sodium, calcium, etc.) to the hydrogen form, producing an acidic effluent. Since acids are much more conductive than mineral salt solutions, the effective sensitivity is increased dramatically. The generally accepted lowest detection limit (LDL) is 0.05 microsiemens per centimeter (μS/cm). With such sensitivity, cation conductivity can be a very useful measurement to detect condenser tube leaks.
B. pH – pH measurement in high purity water can be difficult. There’s an argument pH isn’t a useful measurement, and conductivity is much simpler. A more valid argument is pH can be used in conjunction with conductivity. This is OK if both instruments are calibrated properly and maintained frequently. Otherwise, they can cause confusing “interpretations” of what’s really happening in the process. pH instruments are capable of measuring variances of 0.01 pH unit.
C. Specific Ion Analyses – On-line measurement of specific ions such as sodium and chloride is very effective in detecting cooling water leaks. These instruments can detect ions at levels below that measured by pH or cation conductivity. One manufacturer advertises an LDL of 0.01 ppb for sodium, and 5 ppb for chloride ion.
D. Pneumatics – There are many indirect measurements of condenser tube leakage using vacuum, pressure, etc. These don’t define types or concentrations of actual contaminants.
E. Particle Detection – The development of very sensitive on-line particle counters and particle monitors has provided another tool for leak detection and condensate contamination. Particle monitors that use light “extinction” or “fluctuation” can detect particle concentrations of less than 1 ppb, and particle counters using “laser light obscuration” techniques have a LDL of one particle/ml, or as low as 4 ppt on a volumetric basis (based on particle diameters of 2 microns and above).
Table 1 shows examples of condensate particle counts vs. cation conductivity values based on various cooling water particle concentrations and conductivities. In this situation, based on the respective instrument LDLs, the particle counter would likely indicate a leak prior to the cation conductivity analyzer.
Another feature and possible advantage of particle monitors is that the contaminant does not have to be a solid particle to be detected. For example, oil particles/emulsions will be easily seen at low ppb levels. These substances are not detected by pH, conductivity, ion specific sensors, or pneumatic measurements. However, oils can be very troublesome foulants in condensate polishers, reverse osmosis, etc., and especially if they reach the boiler where they can interfere with water chemistry and cause foaming, carryover, etc.
F. Turbidity – Turbidity has traditionally been a measurement to determine levels of suspended solids. The sensitivity limitations of turbidimeters can be an issue for this sort of application. The typical turbidimeter LDL using conventional 90 degree light scattering (nephelometry) is 0.05 NTU, and particle free water produces 0.03 NTU just from water molecular light scattering. Much scientific investigation using on-line particle counters has verified that a water sample can contain several hundred particles per milliliter at turbidity of 0.06 NTU which is only 0.01 units above the LDL (for example, see Figure 1).
So, due to the small tube leaks that normally occur, and the significant dilution by the condensate flow, turbidity measurement has not been a very effective method for leak detection.
Case History
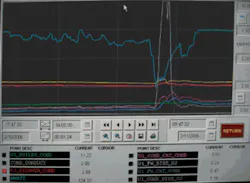
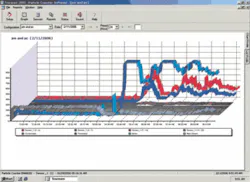
A power plant in the U.S. Southwest installed a particle counter and four particle monitors to track “iron transport” in the boiler cycle (boiler feedwater, steam, condensate, and boiler blowdown). Although condenser leak detection was not part of this study, during the normal course of events the plant experienced a condenser tube leak. This was initially detected by the cation conductivity instruments as indicated on the SCADA monitor (see Figure 1), as well as by data from the particle counter and monitors (see Figure 2). It’s clearly seen that both the cation conductivity and the particle counts increased concurrently. In fact, the magnitude of the increase was much greater for the particle counter. This event confirms that particle detection is a viable method to detect condenser tube leaks.
Conclusion
Although cation conductivity has been the “standard” method for monitoring condenser tube leaks, particle detection instruments should be considered for an alternate or complementary measurement. This is especially true if the cooling water is low in conductivity, and/or high in suspended solids.
About the Author: Bob Bryant is president of Chemtrac Systems Inc., of Norcross, GA. Contact: 770-449-6233, [email protected] or www.chemtrac.com