Dischargers Examining Alternatives for Zero Liquid Discharge
By Kristen Jenkins, Tom Higgins, Jim Mavis, Tom Sandy, Laura Reid, and Ken Martins
There are a number of factors driving industrial dischargers to consider going to zero liquid discharge (ZLD) for their discharges, particularly electric utilities with flue gas desulfurization (FGD) system wastewaters. EPA is developing new effluent limits, which are likely to single out FGD wastewater for stringent standards for metals.
In a recent draft permit for the Public Service of New Hampshire's Merrimack Station, EPA mandated standards based on physical and chemical, followed by biological treatment. In addition, other parameters such as boron, chlorides and total dissolved solids are being considered for regulation, contaminants that are not removed by either physical and chemical treatment or biological treatment. EPA also indicated that while they were not requiring ZLD at this time, they reserved the right to reopen the permit to require it.
Environmental advocacy groups, in suing to overturn new and existing permits, are demanding that coal fired power plant FGD systems go ZLD. In addition, when installing a new FGD system, a treatment system is normally required before the FGD system is completed and wastewater is produced. Wastewaters from different FGD systems vary widely. This, combined with limits becoming more stringent, has made it difficult to provide a treatment system that can achieve current let alone future standards.
Therefore, either because they are facing limits that are not achievable by physical, chemical or biological treatment, or to avoid uncertainty on future limits, companies are evaluating ZLD options. This includes adding ZLD at this time, as well as providing treatment that will achieve current limits, but with a path forward to ZLD without losing investment in current treatment systems.
Approach to ZLD
Like other options, there are multiple paths to achieving ZLD, with widely varying costs and complexity. Usually, the most cost effective approach involves using natural evaporation processes, reducing wastewater flows through operational changes, and taking advantage of potential disposal options for brine in gypsum or fly ash disposal before resorting to the "big gun" of mechanical or thermal evaporation, or crystallization. Taking advantage of naturally occurring evaporation can be low cost. Mechanical or thermal evaporation is more expensive (per gallon). Crystallization is more expensive per gallon and has more technical challenges.
To evaluate options, one first needs a good water and chemical mass balance of the wastewater streams, and potential ways to dispose of brine at the facility. Are gypsum or fly ash disposed at the facility or sold? Water is typically added to fly ash destined for landfilling to prevent dusting and compact the ash to optimum density. Could a brine be substituted for fresh water for this use? How much water is carried with gypsum when disposed? Can the salinity in the absorber be increased to reduce water flow requiring evaporation?
In any event a good water and chemistry balance is needed to evaluate ZLD options.
Chemistry Impacts
One might say that ZLD is not new to electric utilities. There has been extensive experience in evaporation systems to recover water for cooling tower use, in which the predominate salt present is sodium chloride, which is relatively easy to evaporate or crystallize. FGD wastewater tends to be dominated by calcium and magnesium salts of sulfate and chloride, which significantly increase the complexity and costs for evaporation and crystallization processes.
To evaluate alternatives, a rigorous chemistry and mass balance tool is required, accounting for precipitation and species, boiling temperature, vapor pressure, and other physical and chemical interactions taking place at the high temperatures and salinities present in evaporation and crystallization systems.
CH2M HILL has developed and validated such a tool using OLI's Stream Analyzer™ which is a water chemistry model based on an electrolyte thermodynamic framework. The software can be used to solve single point and multiple point survey calculations for trend analysis of temperature, pressure, pH and composition effects, with the ability to mix streams and separate phases. Stream Analyzer predicts phase and species compositional changes for these calculations.
The software was used to evaluate the water chemistry effects, and the results were input to a spreadsheet tool to generate a mass/water balance, size unit processes, and develop the cost of alternative ZLD treatment scenarios. For evaluations with extensive recycle streams, CH2M HILL has developed an approach using Stream Analyzer and a process simulation tool for process modeling, linking to the same spreadsheet tool for sizing and costing. These tools allow the company to evaluate multiple alternatives in a short period of time.
The following provides examples of the use of a water chemistry and mass balance modeling tool to evaluate ZLD alternatives.
Case 1 - ZLD Required
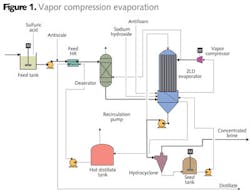
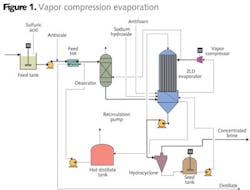
The client had installed a physical and chemical plus biological treatment system for FGD wastewater, but was facing requirements for removal of chlorides from their effluent. CH2M HILL evaluated two options; one in which the wastewater was concentrated and used for fly-ash wetting, and another that evaluated the full crystallization of the wastewater. Option #1: Vapor Compression Evaporation
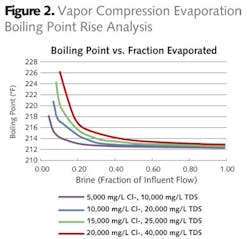
The first option evaluated using vapor compression evaporation (Figure 1) with the resulting brine used for fly ash conditioning prior to landfilling. Vapor compression evaporation is energy efficient, but compression limitations limit the applicability of this technology to a boiling point rise of around 6.5 degrees F. The quality and chlorine content and sulfur of coal impact the quantity and salinity of the FGD wastewater. Therefore, a range of chloride concentrations were evaluated in order to determine the range of brine flows that could be produced by this technology (Figure 2). As salinity increased, the brine factor (volume of brine to initial water) increased, resulting in higher brine flows for the same wastewater flow.
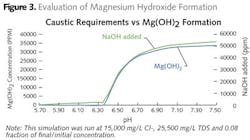
Additional evaporation can be achieved by using steam driven evaporation, when vapor compression evaporation does not result in sufficient volume reduction. Steam evaporation is less efficient than vapor compression, but allows operation at higher boiling points. This higher boiling point comes at a cost of magnesium hydroxide precipitation, which results in significant scaling issues. Magnesium hydroxide formation was not a problem with sodium chloride dominated wastewaters, like cooling tower blowdown.
To prevent magnesium hydroxide formation, magnesium would need to be removed prior to evaporation (softening) of the evaporator operated at a low pH. Magnesium hydroxide formation drops the pH, resulting in increased corrosion on equipment. Sodium hydroxide is added to balance pH for reduced corrosion, while limiting magnesium hydroxide formation. This is illustrated by Figure 3. The optimum pH for this system was 6.3. A higher pH results in excess caustic requirement and magnesium hydroxide scaling. Further concentration of this water would result in higher temperature and increased scaling or lower pH, the latter resulting in greater corrosion potential, requiring more expensive metallurgy.
Evaluation of Magnesium Hydroxide Formation Option #2: Full Crystallization
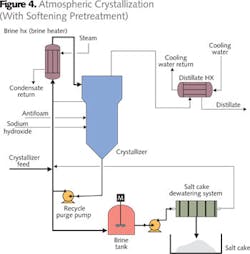
When brine volume cannot be reduced sufficiently, evaporation to a solid (crystallization) is considered. Evaporative concentration produces a brine that is concentrated in calcium and magnesium chloride (sulfate is removed by calcium precipitation in simple evaporation). Unlike sodium chloride, calcium and magnesium chlorides crystallize at unfeasibly high boiling points using atmospheric pressure evaporation equipment. To use atmospheric evaporation, this water must first be softened, replacing the calcium and magnesium with sodium, which then can be crystallized with the chloride (Figure 4). Softening pretreatment requires a huge amount of chemicals and generates a lot of sludge.
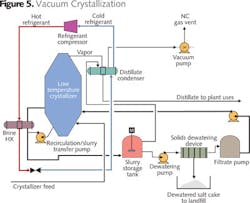
An alternative to softening and atmospheric evaporation/crystallization is vacuum crystallization (Figure 5). Vacuum crystallization generates calcium and magnesium chloride salt which is hygroscopic, and must be carefully managed to reduce exposure to the atmosphere and adsorption of water.
Both options included concentration using vapor compression evaporation as a first step, and had higher capital costs than vapor compression evaporation alone. Therefore, due to the operational issues and higher costs, crystallization would only be selected if an adequate and reliable disposal method for the brine was not available. In this case, ash wetting was determined to be a reliable disposal option. Additional flexibility was provided by reuse of a lined pond for the wastewater allowing flexibility in operation of the evaporator to match availability of fly ash.
FGD Scrubber Blowdown Case 2 - ZLD Considered
In this case, alternatives were evaluated for treatment of FGD Scrubber Blowdown to achieve best available (BAT) treatment limits. Alternatives considered included physical and chemical (Phys-Chem) treatment, biological (for selenium removal) and true ZLD (evaporation and crystallization). These alternatives were evaluated for effectiveness in removing toxic metals, as well as cost-effectiveness and non-cost factors used for BAT determinations.
Fly ash wetting was not available for brine disposal. Therefore, ZLD was based on softening, vapor compression evaporation followed by atmospheric evaporation/crystallization with salt cake disposal.
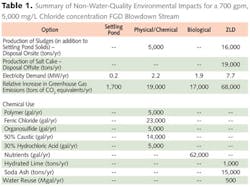
The team evaluated first treating the FGD blowdown with reverse osmosis to reduce the evaporator flows, but found that the little additional concentration was possible with this high salinity waste stream, and initial treatment with RO did not reduce overall costs. Some of the parameters evaluated between the three alternatives are shown in Table 1.
ZLD has the advantage of removing TDS where the other technologies do not. However, when comparing the options based on cost and the amount of toxic materials removed, metals precipitation was much more cost-effective, based on published EPA guidance.
Concluding Thoughts
Zero liquid discharge can be difficult to implement in complex concentrated industrial wastewaters. Due to concentrations of highly soluble components, vapor compression evaporation - which is energy efficient - may not reduce the volume sufficiently to match up with disposal options for this concentrated stream. If this is the case, second stage evaporation or evaporation/crystallization may be required. Increasing brine concentrations to reduce volume can result in a choice between magnesium hydroxide scaling or lowering the pH, which increases corrosion.
If insufficient capacity is available for disposal of concentrated brine, crystallization to form a salt cake must be used. Crystallization of complex wastewaters can produce salts that are difficult to handle such as hygroscopic salts like calcium and magnesium chloride or formate based.
ZLD for industrial wastewater requires evaluation of complex water chemistry to evaluate operating parameters like boiling point rise, pH, and solids formed, and a chemistry model is needed given the wide range of constituents and potentially high concentrations. ZLD is most applicable where there is a disposal option for concentrated brine, regulations require removal of total dissolved solids, or there is no option for disposal of the wastewater stream.
About the Authors: The authors all work for CH2M Hill. Kristen Jenkins is Industrial Wastewater Global Technology Lead, Principal Technologist; Thomas E. Higgins is vice president and technology fellow; A. Thomas Sandy is Industrial Wastewater Service Team Leader; Jim Mavis is Principal Technologist - Chemical Processes; and Ken Martins is Senior Principal Process Engineer. Laura Reid is an Associate Engineer.