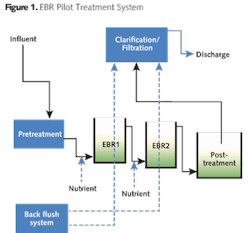
Pilot system successfully removes metals, inorganics
By A. Opara, D.J. Adams, and M. Peoples
An underground mining operation producing copper, lead, zinc, silver and gold uses three floatation circuits for metal recovery. Suppressants (e.g., cyanides) and collectors (e.g., xanthates) are added at each step to recover only the desired products. Water and solids from the floatation circuit are discharged to a tailing pond, where solids are allowed to settle and water is recirculated to the circuits.
Constant water reuse at the mining operation, without treatment, has resulted in declining water quality. The polluted water cannot be discharged to the environment and has resulted in significantly reduced floatation recoveries. The concentration of metals (i.e., selenium, antimony, cadmium, copper, lead, molybdenum, silver, and zinc) and chemical reagents, such as collectors and suppressants, is increasing over time. High concentration of complex organic compounds and other contaminants (over 700 mg/L COD; TDS of over 4,000 mg/L; selenium 2.0-3.5 mg/L) renders this wastewater difficult to treat using conventional biological, chemical and/or physical processes.
EBR Technology
In an attempt to find a suitable water treatment system, the mine operators decided to pilot test Electro-Biochemical Reactor (EBR) technology, which can improve upon conventional biological treatment methods and is effective in the removal of metals (e.g., selenium, arsenic, mercury, cadmium) and inorganic contaminants from wastewaters and mine process waters.
All biological and chemical metal reduction/removal methods are based on redox reactions. Electrons are needed for contaminant transformations, and rely on chemicals and nutrients to provide these electrons. Using selenium as an example, in the EBR biological treatment system the selenium oxidation state is not as crucial as it would be in chemical and some other treatment processes. Selenate is reduced to selenite, which in turn is reduced to elemental selenium, which is virtually insoluble.
The EBR overcomes the shortcomings of conventional bioreactors by directly supplying excess electrons to the reactor and microbes, using a low applied voltage: 1 to 3 volts is all that is required for the EBR (1-volt supplies approximately 1 trillion, trillion electrons). These electrons replace the electrons normally supplied by excess nutrients and chemicals, at a considerable savings. The provided electrons make the reactors more controllable, economical, and robust than past generations of biological treatment systems. Moreover, they provide readily available electrons for microbial growth and contaminant removal, resulting in better performance in less time and space and with greater efficiency.
Pilot System
The PLC-controlled pilot system is completely contained and designed for flow rates of 0.5 to 3 LPM.
Experience had shown that conventional selenium removal methods including the EPA’s best demonstrated available technology (BDAT) — iron co-precipitation — had not been able to meet the discharge limit (0.02 mg/L). The EBR pilot system removed all examined metals and inorganics to below discharge criteria; >93% to >99% removals were obtained.
All metals, except for molybdenum, were present in the influent wastewaters at concentrations above the allowable discharge limit. Selenium was removed from influent values averaging 2.73 mg/L to an average 0.002 mg/L in the system effluent. Discharge criteria were met within 16 hours in the first EBR treatment stage.
Significant changes in influent selenium concentration and temperature had no effect on EBR system performance. Nutrient costs were significantly decreased through conversion and use of toxic floatation organics in the EBR process.
Conclusions
- The EBR technology successfully removed high selenium concentrations (2.0-3.5 mg/L) from difficult floatation-affected wastewaters to discharge criteria using short HRTs and to ultra-low levels (≤ 0.002 mg/L) using longer HRTs.
• Selenium and other metals are precipitated in the EBR system in stable elemental and sulfide forms, significantly reducing the sludge volume and providing a potential for recovery at the end of reactor life.
- Reduction of various other metals occurred concurrently with selenium reduction:
• Antimony, cadmium, copper, lead, molybdenum, silver, and zinc.
- In addition to metals removal, various inorganic species were removed by the EBRs:
• Nitrate-N, nitrite-N, WAD cyanide, total cyanide.
- Preliminary metallurgical tests using EBR effluent waters showed that treated waters may significantly increase the floatation recovery of precious and base metals.
- The initial EBR evaluation indicates over 25% lower capital costs and up to 50% lower operational costs than conventional bioreactors and other treatment methods.
- The EBR system produces more controllable and stable bioreactor environments.
- Power requirements for a full-scale facility can be supplied by a small solar grid, because of the low DC voltage potential needed for the EBRs.