Performance Expectation of Ion Exchange Resins Under Variable Operating Conditions in a Makeup Demineralizer
By Terrence Heller
The objective of optimizing demineralizer systems is ultimately to reduce operating cost by minimizing regenerant and maximizing throughput while meeting your plant's water quality requirements. Achieving this balance is desirable but choosing the appropriate resin and system configuration can be complicated. Modifying existing systems to observe change is difficult and can result in unexpected drawbacks.
One alternative is to use a computer simulation program to predict the performance of system changes. The Purolite Company offers a computer design program, PureDesign, which models existing systems for the purpose of observing the benefit of change. System performance will vary from theoretical calculations but is effectively related.
The PureDesign program was used recently to study the impact of changes to a treatment system at a southern manufacturing plant with three demineralizer trains. Each train has a strong acid cation vessel that holds 466 cubic feet of resin and an anion vessel that holds 255 cubic feet of resin.
There is no degasifier. Each train is designed to flow at 622 gpm. The water source is a medium sized river with an ionic loading averaging 83 ppm TDS, a Total Na/Total Cation of 18%, Total SiO2/Total Anion of 4%, and a TOC of .20 ppm.
Since individual demineralizers will perform differently from location to location, the comparison of both the system and resin performance is based on the water quality in Table 1 unless otherwise specified.
Process Options
Cation Resin - Strong acid cations most commonly used for demineralization are 8% and 10% cross-linked styrenic gels. Both these resins have similar performance even though the 10% cation has higher total capacity.
This similar performance is due to slower kinetics of the 10% gel, which reduces operating capacity and regeneration efficiency to that of the 8% gel. Advantage of the 10% is its longer life especially if cations are subject to oxidizing chemicals.
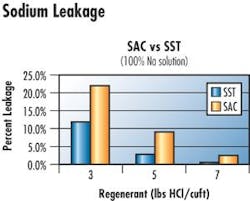
A relatively new surface-functionalized SAC styrenic gel product (SST or Shallow Shell Technology) has been receiving attention for use in demineralization because of low sodium leakage.
Aspects evaluated for these three cation resins included operating capacity and sodium leakage when regenerated with hydrochloric acid (HCl) and sulfuric acid (H2SO4). Performance of the 8% cation operated in a co-flow and counter-flow regenerated systems was included because of features this technology offers.
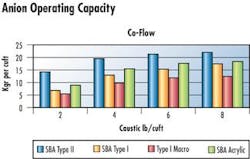
Anion Resin - Commonly used SBA's include styrenic Type I gel, Type II gel, and Acrylic Type I gels resins.
Macroporous resins have advantages where there are chemical, mechanical and osmotic stresses so a Type I macroporous anion was included. These anions were evaluated for operating capacity and silica leakage.
Performance of each was compared in co-flow and counter-flow regenerated systems. Regeneration temperature was set according to the anion's tolerance to temperature. Type I gel and macroporous resins are regenerated at 120°F. Type II and Acrylic anions are regenerated at 95°F.
Systems - Co-flow and counter-flow regenerated systems were compared to demonstrate performance differences in throughput, leakage and regenerant usage. The most difficult aspect in discussing counter-flow technology is the variations available between co-flow and true counter- flow regenerated systems.
This discussion compares only co-flow regenerated systems that have service flows from the top of the vessel down and regenerant flow also from the top down. Counter-flow systems have service from the bottom of the vessel upward discharging from the top creating a packed resin polishing zone and a fluidized resin zone. Regenerant, though, enters from the top flowing down through the bed exiting the bottoms.
Optimization
In this specific southern manufacturing plant, the PureDesign modeling program determined that maximizing throughput and minimizing regenerant use while maintaining appropriate effluent quality was best achieved in a counter-flow regeneration system. The preferred resins would be SAC gel (SST if ultra low leakage is required) regenerated with 4 lbs HCl and Type II gel anion regenerated with 4 lbs NaOH. The software predicted this program would achieved all objectives with a 25% throughput increase; a reduced acid consumption by 55% and caustic by 50%, while maintaining silica leakage at 10 ppb and Na at 11 ppb.
Changing regeneration mode of an existing plant is expensive, as is switching from H2SO4 to HCl. Therefore another option considered was operating the system as designed, assuming co-flow regeneration with H2S04. In that case, computer modeling indicated the best cation choice would be SST regenerated at 6 lbs H2S04 in a 2%- 4% or 6% stepwise regeneration. This is advantageous if influent calcium is present. The economically preferred anion was Type II gel regenerated with 6 lbs NaOH/cu ft.
According to the simulation, throughput would increase by 36% compared to Type I anion. Regenerant consumption would remain constant per regeneration but savings would be due to 36% fewer regenerations. Conductivity of the effluent water would decrease by 50%, but silica would increase from 10 ppb to 33 ppb. Regeneration temperature would need to be reduced to 95 degrees F, saving some in terms of energy. Polishers following the single bed vessels would remove excess SiO2 and may have a greater throughput especially if conductivity is the limiting factor.
The option to use standard 8% or 10% SAC regenerated with sulfuric acid at 6 lb/cu ft and Type I anion resin regenerated with 6 lbs/cu ft caustic would produce an effluent conductivity of 2 uS/cm and sodium leakage of 203 ppb, according to the modeling program. Throughput would be 709 million gallons and silica leakage will be 12 ppb.
This combination of resin is the most commonly chosen because of lower initial cost for both resin products and low silica leakage from the anion. Also, Type I anion resin would have a longer life than Type II anion. What is not considered here is the higher operating cost, which quickly surpasses initial costs and operational benefits of the SST and Type II.
In situations where TOC is a limiting factor for anion throughput and life, an acrylic anion may be used in place of Type I or Type II anions.
In this particular case, modeling indicated acrylic resins would tolerate three times the organic load of styrenic resins before regeneration. Additionally, throughput would be increased 14%, regenerant requirement would again remain constant per regeneration but there would be less regeneration, and silica leakage would increase to 19 ppb.
If organic levels are excessive, use of weakly basic resins or an organic trap may also be considered. Otherwise ionic loading should be secondary to organic loading and shorter runs programmed to reduce TOC buildup. Acrylic anions will generally be more expensive than styrenic anions. Notable difficulty with acrylics is their long rinse requirement with age.
About the author: Terrence Heller is South East Regional Sales Manager for the Purolite Company. He has an MS degree from Central Michigan University in Environmental Science and has worked for 12 years in the field of ion exchange. Earlier experience was with water treatment and wastewater treatment with Grace Dearborn. This article was adapted from a paper presented at the 63rd Annual International Water Conference.
About the International Water Conference: The International Water Conference is the world's premier Conference on Industrial Water Treatment. IWC has been conducted by the Engineers' Society of Western Pennsylvania (ESWP) since 1940. IWC is attended by the leading experts, engineers and academics in the USA, Canada and Europe who are actively pursuing advancements in industrial water treatment and processing technology . The Conference and Exhibition is held annually in October, in Pittsburgh PA. For continually upgraded agenda, and registration information, visit eswp.com/water.html.