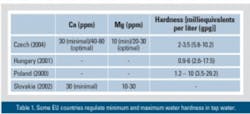
Tap water hardness may vary widely in different regions and countries from ~1.45 to ~81.75 grains per gallon (gpg). More often, tap water is relatively hard (more than 6 gpg). For example, about two-thirds of the consumers in Estonia have tap water with hardness between 6 and 15 gpg. Part 1 of this two-part series discusses the hardness requirements for tap water and how reverse osmosis (RO) treatment can help remove minerals in addition to toxins and other impurities.
Optimum mineral content
Opinions differ on the optimal mineral content of drinking water. Heating hard water produces scale, which destroys heating elements. Soft water is recommended for the preparation of most food and drinks. At the same time, calcium (Ca), magnesium (Mg) and otherOne opinion is that drinking water can be free of all minerals because sufficient amounts of these minerals are present in milk-based and other products. Another view is that drinking water should contain a certain amount of these minerals (Ca, Mg, carbonates, among others) to assist normal bowel movement and other bodily functions.
Reference 1 suggests an inversely proportional dependence exists between water hardness and death statistics from heart illnesses. The Mg concentration was found to be especially critical (twice that of the Ca influence). The same study also noted that deaths caused by heart failure depend on the concentration of certain toxic impurities (Mn, Cu).1
In 2011, the World Health Organization (WHO) made an official recommendation based on long-term medical studies in France and other countries: "Combined Mg and Ca water hardness should be 4.38 gpg or more." It was noted that the Mg concentration should equal or exceed 1.45 gpg. Many bottled water manufacturers, including some leading international brands, adjusted the mineral content of their water to include more magnesium bicarbonate after the recommendations were published.2An average consumer uses some water for drinking, but even more water for making drinks including tea and coffee and for cooking and baking. Soft water is best for these purposes because it has better water-based extractions from meat, grains of coffee and cereals, and yeast functions much better during the fermentation processes. Soft water also significantly improves the taste of these products. Water with a hardness of less than 1.45 gpg is best for making coffee, tea and soup and to ferment dough for baking bread. Professional coffee makers require small concentrations of Mg for the optimal coffee extraction, taste and appearance (such as the foam on some coffee drinks). Many popular beers require water that is practically demineralized (see Table 2).3
Considering that the majority of consumers at home use the same water for drinking and for cooking, a compromise of 6 ppm Mg concentration, between the WHO recommendation and small mineralization for cooking (1.45 to 1.75 gpg or less), is acceptable.
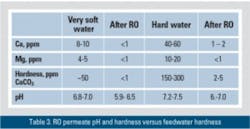
Among widely used home water purifiers, RO membranes are the most efficient for removing toxic compounds and other impurities. At the same time, RO membranes typically reduce water hardness from 95 to 99.9 percent. The percent reduction depends on the selectivity of the RO membrane and the degree of the permeate recovery. The permeate pH typically varies between 5.9 and 7.0 (very hard water).
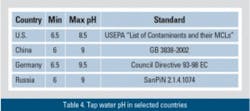
Tap water pH is regulated to between 6 to 6.5 and 8.5 to 9.5 depending on the country (see Table 4).
Part 2 is featured in the December issue. It discusses how water in an RO filter without flow increases in mineral content.
References- Schroeder, Henry A., MD. (1966, Jan. 10). Municipal Drinking Water and Cardiovascular Death Rates. Jama, Vol. 195, No. 2, pp. 125-129.
- Kozisek, F. (2005) Health risks from drinking demineralized water. In Nutrients in Drinking Water, pp. 148-163, World Health Organization, Geneva. From who.int/water_sanitation_health/dwq/nutrientsindw/en/index.html
- "Treating water to brew the perfect cup of coffee." (2014, Dec.). Water Technology. From watertechonline.com/bottled-water/
treating-water-to-brew-the-perfect-cup-of-coffee/
Alexey Aksenov is senior research chemist of WestAqua Ou, Aquaphor Group, Narva Estonia. He is a graduate of the St.Petersburg State Institute of Technology Electrochemistry department. He may be reached at [email protected].
Alexander Mitilineo is head of R&D Chemistry Dept. of WestAqua Ou, Aquaphor Group, Narva Estonia. He is a graduate of the St. Peterburg State University Inorganic chemistry department. He may be reached at [email protected].
Alexander Polyakov is vice president, technical sales of WestAqua Ou, Aquaphor Group, Narva Estonia. He is a graduate of the Leningrad State University Physical Chemistry Department’s Ph.D. program. He may be reached at [email protected].
Joseph L. Shmidt, CEO of Aquaphor Group, was also co-founder of Membrex Inc. (now a part of Osmonics, GE Water). He earned his Ph.D. and M.S. degrees in chemical engineering from Columbia University. He may be reached at [email protected].