Production of high-quality demineralized water for steam generation units
Demineralized water is a raw material necessary for generating steam in boilers. This steam is used to run turbines or as a raw material for manufacturing. Creating demineralized water is a thorough process. In this article, we will cover the basic demineralization process as well as causes and fixes for some common unit disturbances.
Potable water
Potable water is received by a feed potable water tank and metered by a utility.
Chemical Specification of Potable Water
| Temperature | Ambient |
| pH ( at 25o C) | Range about [8 to 8.5] pH |
| Chlorine | Range about [0.5 to 1] mg/liter |
| Conductivity | Range about [80 to 160] µs/cm |
| Total Dissolved Solids (asCaCO3) | Range about [40 to 80] mg/liter |
Demineralized water
A demineralized water facility produces process water and boiler feed water for use in steam generation by boilers. It also returns steam condensate from plants and pretreats it. A demineralized water facility consists of the following:
- • Demineralized raw water tanks and raw demineralized water pumps
- • Demineralize unit
- • Demineralized water tanks
- • Demineralized water distribution pumps
- • Return condensate tanks
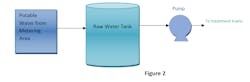
Raw water tanks and raw water pumps
A raw water tank is used to feed water to a treatment water train and produce demineralized water.
Activated carbon filter
Raw water, consisting of potable water, treated condensate and recovery water is pumped into an activated carbon filter (ACF). These filters reduce free chlorine below 0.1mg/l and remove some of the suspended solids.
Cation exchanger
A cation exchanger is used to displace positive ions by hydrogen ions. R-H resin converts salts to their respective acids, like calcium carbonate to carbonic acid, sodium chloride to hydrochloric acid, and sulfates into sulfuric acid.
Because of acids, the water passing out of cation exchanger resin becomes acidic, with pH levels between 2 and 4.
Degasifier
The acidic water passing out of the cation exchanger flows to a degasifier tower. In this tower, carbonic acid is stripped by the fresh air blown upward by a fan from the bottom of the packing. The air containing CO2 is exhausted through the top opening of the degasifier.
Anion exchanger
An anion exchanger is used to displace negative ions by hydroxide ion. R-OH resin absorbs acids.
Mixed bed polisher
Most of the ions, including silica, are removed by the cation exchanger, anion exchanger and degasifier. To produce the best quality treated water, it is passed through a mixed bed polisher, which contains both R-H and R-OH resins. The polisher has a capacity of 270 m3/h (typically per train).
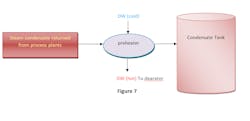
Condensate pretreatment and condensate tanks
Steam condensate returned from process plants is cooled by a demineralized water preheater. It is then stored in a condensate tank. Condensate from this tank is pumped into a cation exchange filter (CEF). The CEF is used to remove suspended solids, mostly iron oxides, which are removed more effectively by cation resin and then routed to the demineralized raw water tanks.
Regeneration
Ion exchange resin loses its ion exchange ability during service. It can also be exhausted because of nonionic impurities, suspended solids, colloidal matter and dissolved solids. Therefore, regeneration operation, including backwashing, is required. When the quality of the treated water becomes bad or the quantity of the treated water reaches the set value, the service operation should be stopped. And the regeneration operation should be started.
- Regeneration Device
The regeneration device consists of hydrochloric acid (HCL) and sodium hydroxide (NaOH) storage vessels, measuring vessels and ejectors to dose the chemical. It also includes a regeneration pump system to provide the diluted water of the chemicals. Regeneration devices are installed near demineralized water units, except regeneration pumps, where they are protected by the bank of acid-proofed concrete.
The HCL and NaOH storage vessels are filled by tank lorry, and each chemical is transferred to the measuring vessel by gravity. The regeneration device pumps demineralized water to both HCL and NaOH measuring vessels for dilution.
To remove silica from an anion exchanger, a warm caustic soda solution is needed. This is produced by using a steam silencer ejector.
-Normal regeneration
Normal regeneration is carried out by using HCL acid for the cation exchanger and NaOH for the anion exchanger with water. A surface wash of the resin bed is performed to maintain the advantage of counter current regeneration (CCR).
- Special backwashing (double regeneration)
- Regeneration steps
1- Cation Exchanger
2-Anion Exchanger
Weak anion resin
Strong anion resin
Demineralized Water Tanks and Pumps
Demineralized water tanks are used to store demineralized water and supply boiler feed water to boilers.
Neutralization System
Acid and alkaline waste produced during the regeneration and blowdown liquid from steam facilities are routed to the neutralization pond where waste is mixed, neutralized and discharged to the seawater facilities. By neutralizing, water circulation and air mixing start first. The water circulation continues until the waste is discharged. The pH of the wastewater is always checked and indicated by a pH controller, as either acidic or alkali.
Caustic soda is injected by gravity from a caustic soda vessel because regeneration wastewater of demineralized water is normally acidic. The required pH value for wastewater in a neutralization pond before discharge ranges between 6 and 9 pH.
Recovering system
The recovery pond recovers wastewater, which has a good quality that can be used as a source for demineralizer. Wastewater taken from the recovery pond comes from:
• Surface wash water of the cation and anion exchangers, which is sent from the recovery pond to a raw water tank.
• Backwash, purge and rinse water of the mixed bed polisher, which is sent from the recovery pond to a demineralized raw water tank.
• Backwash water of the activated carbon filter, which is recovered to the condensate tank.
Dearator Preheater
Dearator feed water is pumped to the dearators through the preheater. The dearator preheater heats the water to approximately 80oC by exchanging heat with condensate from the process plants. The temperature is measured before and after the dearator preheater.
Steam condensate passes through the shell side with an operating pressure of 3 kg/cm2G, and an operating temperature about 133oC to 50oC. Demineralized water passes through the tube side with an operating pressure of 7.5 kg/cm2G and operating temperature about 45oC to 84.1oC.
Demine System problems and troubleshooting
Chemical (resin oxidation)
Resins are composed of crosslinked polymers that can stand up to a variety of substances. Still, they are vulnerable to oxidizing agents, such as nitrates, peroxides, halogen compounds, chlorine and hypochlorite compounds, among others. When present in a feed stream, oxidants degrade IX resin polymers, causing them to deform and compact over time. This compaction obstructs the flow of liquids through the resin bed. Resin oxidation problems can be avoided by treating raw water with activated carbon filters before cation exchange and by monitoring raw water quality periodically. Also, operators can apply a reducing agent when oxidizing agents are observed.
Thermal resin degradation
Thermal degradation alters the resin’s molecular structure such that it is no longer able to bind with the functional groups of ions. Operators can save resin from thermal degradation by considering inlet temperature conditions into the initial design stage, which is normally atmospheric temperature. Also, they can use an online temperature sensor in the feed line of the demineralized water unit. Data sheets from resin vendors describe the resin’s ability to withstand the range of operating conditions without degradation.
Mechanical and operational
Channeling occurs when liquids pass through the resin unevenly, carving pathways that result in the uneven exhaustion of the resin, and breakthrough of untreated solution into the effluent stream. During normal operation operators can avoid channeling by adjusting incorrect flow rates or maintaining adequate backwashing. If operation troubleshooting does not prevent channeling, try repairing failures in the distributor mechanism.
Inadequate regeneration can result when chemical solutions are applied incorrectly. Facilities can keep adequate regeneration by following the resin manufacturer’s guidelines for chemical concentration, application time and flow control. This will be done through checklists and standard operating manual instructions.
Resin loss migration occurs when resin beads flow out of a column, or flow from one vessel to another. There are multiple causes for resin loss, including excessive backwashing and mechanical failures in drain screening or other resin retention equipment. Resin loss may also result from fragmentation of resin beads due to exposure to high temperatures, chlorine and/or osmotic shock, allowing the resin particles to pass through even intact retention screens. Resin loss and migration reduces overall system capacity and efficiency. In demineralization systems, for example, the migration of cation resin into the anion unit can result in sodium leakage and excess rinse time. To prevent the loss of resin, avoid excessive backwashing by adjusting the flow. Another way to prevent loss of resin would be to repair mechanical failures for drain screens or other resin retention equipment during maintenance periods. As with many operations, preventive maintenance is key method.
The most common problems are mechanical failures like support collapse or resin leakage and chemical oxidation for resin. Thermal degradation can happen, but it occurs rarely.
Conclusion
Demineralized water is necessary to avoid the type of scale accumulation in boiler or heat exchanger tubes that would occur with typical potable water. To produce the appropriate demineralized water, plant managers must understand the fundamental steps covered in this article. Otherwise, they run the risk of complications such as thermal degradation or chemical and mechanical failures. As with most operations, steam generation units can be run safely and effectively by following all operation manuals, checklist instructions and troubleshooting protocols.