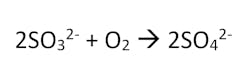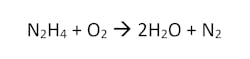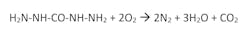Boiler corrosion: Mechanical and chemical methods for control

Industrial boilers play a foundational role in the operation of modern society. From heating to chemical manufacturing to electric power generation, there is no questioning the importance steam production plays in the creation of a vast amount of goods we all use.
While this process of steam production is relatively straightforward (an external source of heat energy applied to water, either directly or indirectly), the secondary processes involved must be monitored with just as much criticality. Items such as water pre-treatment and boiler internal treatment, fuel pre-treatment, flue gas after-treatment and mechanical maintenance all play a role in the long-term reliable operation of boilers. Within the water treatment segment of boiler operation, two main mechanisms affect their efficiency and longevity – scale/deposition and corrosion. While the former typically is a result of inadequate or improper pre-treatment, the latter is typically a result of improper internal chemistry treatment and/or associated mechanical equipment. This article will focus on the corrosion aspect of boiler operation, and methods for mitigation and monitoring.
An explanation of corrosion and its effects
Corrosion is the gradual deterioration and loss of a material by electrochemical reactions with its environment; a natural process that converts a refined metal into a more stable oxide. A corrosion cell has two components: the cathode and the anode. The cathodic reaction is a reduction of oxygen to the hydroxide ion at the surface of the metal, and the anodic reaction is the oxidization of the metal into soluble ions. Co-occurring, these combined reactions result in a net loss of base metal. Throughout a boiler system, the primary cause of corrosion is the presence of dissolved oxygen in the water, causing a reaction that forms iron oxide (rust).
Several different characteristics of the water determine the corrosion rate: dissolved oxygen, pH and temperature all affect corrosion severity and speed. Different corrosive properties threaten the various stages throughout a boiler system. In the feedwater and boiler, dissolved oxygen is the perpetrator of corrosion. Water will absorb oxygen directly from the atmosphere and introduce it to the feedwater tank through makeup. However, unnecessary turbulence can increase the absorption of oxygen in makeup water.
In the condensate piping, corrosion is mainly caused by the formation of carbonic acid. When the condensate travels back to the feedwater, CO2 (carbon dioxide) absorbs into the water and forms H2CO3 (carbonic acid). With such high-purity water in the condensate, it does not take much dissolved CO2 to lower the pH to a corrosive range. Higher makeup alkalinity will increase the likelihood of this, making a condensate pipe more susceptible to carbonic acid attack. This kind of corrosion will cause grooving on the condensate piping.
As corrosion occurs, material released from the anode will deposit on any metal surface. Iron deposition will attach to and effectively insulate the tubes in a boiler. This requires the boiler to fire harder to achieve the same heat exchange, which reduces the boiler's efficiency. This can even cause the boiler tubes to overheat and lose structural integrity. Deposits can come from corrosion within the boiler or be pumped in from corrosion in the feedwater.
On the other hand, if corrosion occurs in the boiler, different areas on the tubes or wall will have a material loss, which can cause, in severe cases, a tube leak or failure, particularly with high-pressure watertube boilers. Metal loss in the feedwater does not have adverse performance effects; however, it can affect the boiler, as stated above. Pitting and other types of corrosion in the feedwater tank can also indicate the boiler's health.
Corrosion management — mechanical oxygen removal
There are several ways to minimize corrosion in a boiler system and protect the metal surfaces throughout. The first step to reducing dissolved oxygen should be through mechanical means. The most effective way to do this is with a deaerator. This piece of equipment uses steam to remove non-condensable gases from makeup and condensate streams. As the source water is sprayed into the vessel, steam flows cross or counter to the water flow. The water is heated to the full saturation temperature corresponding to the steam pressure in the deaerator and causes the gases to have reduced solubility and be vented out to atmosphere. The resultant degasified water then falls into the storage tank area below the deaerator.
A properly functioning deaerator can remove most dissolved gases without the help of chemical treatment. It is crucial to monitor the performance of the tank by periodically checking the pressure of the deaerator and the temperature of the water in the storage tank. The storage tank temperature should be within 2°F of the steam saturation temperature corresponding to the pressure indication. If the tank temperature is more than 2°F lower, that indicates poor mixing of the steam and water — typically indicative of malfunctioning spray nozzles. Additionally, the vent temperatures should be checked — if the deaerator has multiple vents, the temperature should be within 1°F of each other, and the temperature should be 3-4°F lower than the saturation temperature.
If a deaerator is not an option, there are several ways to remove dissolved gases in a standard feedwater tank. However, chemical treatment may be necessary downstream depending on the dissolved oxygen in the source water. Steam sparging is a common method of preheating the feedwater and removing the dissolved oxygen. Using steam off the boiler, raising the temperature of the feedwater will reduce the solubility of dissolved oxygen. The heating of the feedwater tank is a balancing act — ideally, the feedwater temperature would be the saturation temperature at the given atmospheric pressure where it is located. Unfortunately, this leads to feedwater pump cavitation issues. Typically, 10°-15°F below saturation temperature is the maximum safe range, resulting in approximately 2 ppm of dissolved oxygen. Other options, such as electric immersion heating elements, can be used to achieve this same effect. If a process allows, increasing the condensate return will also help increase temperature. However, some industries, like feed production, do not have enough condensate supply to heat the makeup water.
Corrosion management — chemical oxygen removal
While mechanical equipment are a very effective means to initially mitigate corrosion in pre-boiler and boiler circuitry, they do have their limitations. A feedwater tank, in its limited ability to remove dissolved oxygen, does need supplemental means to chemically remove the remainder. A properly functioning deaerator does not need much assistance chemically to remove dissolved oxygen downstream, but in a power boiler configuration there is a condensate circuit that can be subjected to vacuum conditions and subsequent oxygen intrusion. In systems such as these, a chemical oxygen scavenger must be injected to properly passivate this section, given that the system contains copper alloy heat exchangers.
Depending on the boiler pressure and steam use, there will be a preferred oxygen scavenger/passivator product that should be used. At times, there are semantics involved in the nomenclature of these chemicals (oxygen scavenger vs. passivator), but the root chemical reaction involved here is the chemical with dissolved oxygen, in order to produce a sufficiently reducing atmosphere to promote the formation of a protective magnetite (Fe3O4) barrier on the surface of steel piping, and protective cuprous oxide (Cu2O) on the surfaces of copper tubing. This is not an all-inclusive list of scavengers, but rather a highlight of the most commonly used ones.
Sulfite
Sulfite, in the form of either sodium sulfite, sodium bisulfite or sodium metabisulfite, is the most commonly used oxygen scavenger in low pressure boilers, primarily due to cost. Sulfite reacts with dissolved oxygen to form sulfate, in the following basic equation:
While sulfite is quite effective, particularly with the use of catalysts like cobalt salts, it does have limitations. It should not be used in boilers above 900 PSI, as sulfite can decompose into sulfur dioxide (SO2) and hydrogen sulfide (H2S) gases, both corrosive, which can then carry off with the steam and cause corrosion issues in the condensate return circuit. Additionally, sulfite cannot be used in boiler systems that use feedwater for steam attemporation, as this will introduce solids into the steam and cause deposition issues on downstream equipment.
Hydrazine
Hydrazine is a simple, volatile, oxygen scavenger that does not contribute to dissolved solids in its reaction with dissolved oxygen. In this sense, it can be used without issue in high pressure boilers utilizing attemporation. The reaction is as follows:
The reaction proceeds slowly at lower temperatures, so it must be fed at high dosages if being used to treat a steam condenser hotwell. At temperatures above 400°F, hydrazine decomposes to form ammonia. With a system containing copper, this can contribute to corrosion. Additionally, hydrazine is a suspected carcinogen, and special procedures must be in place for proper handling in order to mitigate exposure. Hydrazine is seldom used other than in nuclear steam turbine cycles.
Carbohydrazide
Developed as an alternate to hydrazine without the shortcomings, carbohydrazide has the added benefit of reacting with dissolved oxygen significantly faster at lower temperatures. The reaction is as follows:
It should be noted that not all boiler systems are required to have a chemical oxygen scavenger. High-purity power boilers without copper operate with better corrosion mitigation utilizing chemistry programs that either allow oxygen ingress up to 20 ppb (All-Volatile Treatment — Oxidizing) or inject gaseous oxygen to maintain 30-50 ppb (Oxygenated Treatment) in the feedwater.
Corrosion management — neutralizing amines
Carbon steel must be in contact with water in an alkaline pH range in order to minimize corrosion. The absolute pH values, and ranges of values, differs based on the water purity and whether the system also contains copper alloy (e.g. feedwater heaters or condensers). For example, a low-pressure firetube boiler using softened water makeup should have a feedwater pH in the range of 8.3-10.5 (per ASME Guidelines), while a high-pressure boiler operating with deionized water makeup should have a feedwater pH in the range of 9.0-9.3 (with copper in system) or as high as 9.6-10.0 (no copper), per EPRI guidelines. In order to achieve proper alkalinity in the feedwater as well as the condensate circuits, the addition of a volatile amine is needed. There are myriad products in the water treatment market intended for this purpose — from as simple as ammonia to complex amines such as cyclohexylamine and morpholine. The proper selection of an amine lies in matching its properties (namely basicity, volatility and thermal stability) with the system in which its use is intended.
- Basicity is the ability of the amine to increase pH. Morpholine has a practical upper pH limit of 9.2, while ammonia can increase pH much further.
- Volatility is the ability of the amine to carry off with the steam from the boiler and condition the condensate. For some amines, it can be highly dependent on the operating pressure of the boiler. For example, cyclohexylamine is four times more volatile at 600 PSI as compared to 10 PSI, whereas ammonia stays relatively unchanged through that same pressure range.
- Thermal stability refers to the ability of an amine to resist decomposition into smaller organic molecules — typically smaller amines or organic acids. Organic acids are especially impactful negatively in high-purity system, where they contribute significantly to conductivity, lower pH and increase corrosion. Typically, larger amines are less thermally stable than smaller amines.
Corrosion monitoring — key performance indicators
Even with all industry treatment guidelines being followed, there still needs to be feedback observed in order to determine the optimized point at which to operate in the guidelines. A feedwater pH guideline of 8.3-10.0 is quite wide, and there will be an optimized point within that range for a given boiler. This observed feedback comes in the form of corrosion monitoring (at minimum iron, and copper for those systems which contain it). This can more specifically be divided into spot-testing or long-term accumulated sample testing. In all instances, the testing involves bench spectrophotographic methods (using a spectrometer or colorimeter, or Atomic Absorption/Atomic Emission Spectroscopy) and comparing them to industry standards.
Spot-testing involves taking a grab sample (feedwater, condensate, etc.) under a known operating condition. The conditions (steam load, pressure, etc.) should be tracked along with the resultant iron and/or copper concentrations in the sample. Long-term accumulated sampling involves running a particular sample through a piece of equipment (ion exchange cartridge or corrosion product sampler with combined particulate filter and ion exchange pad) over an extended period of time. At the end of a designated time period, the total volume of sample throughput is recorded along with the metal analysis result from that collected, in order to calculate a concentration value. Typically, low pressure/low purity water boiler metal transport values are tested in parts per million (PPM) concentration, and high pressure/high purity boiler transport values are tested in parts per billion (PPB) concentration.
Spot-testing pros
- 100% certainty of metal transport value under the observed operating conditions.
- Patterns can be detected in transport based on changes in conditions.
Spot-testing cons
- Pattern detection requires large numbers of samples over time.
- High purity boiler iron testing requires extremely clean testing/lab conditions in order to provide accurate, repeatable results.
Long-term accumulated sample testing pros
- Requires less labor, as there are fewer samples to test.
- Gives good general operating condition assessment of boiler system.
Long-term accumulated sample testing cons
- Sample analysis generally requires specialized laboratory.
- Results only show an average value over time. No correlation to specific mechanical/ external events can be derived.
With modern industry having such reliance on boilers and steam production for critical infrastructure items, it is paramount for boiler owners, operators and vendors alike to focus on reliability, efficiency and longevity. Boiler corrosion is one significant aspect that can affect these three performance characteristics, and as such is something that should be monitored and controlled using industry best practice guidelines (either ASME or EPRI, depending on boiler type). The relatively small amount of capital and labor costs needed for corrosion control will be miniscule in comparison to large-scale equipment damage and subsequent replacement.
Kurt Bayburt is the Technical Director for H2 Global Solutions. He has 20 years of industrial water treatment experience, both in sales and end-user roles.
Silas Norwood is the Operations Manager at H2 Global Solutions.
About the Author
Kurt Bayburt
Technical Director for H2 Global Solutions
Kurt Bayburt is the Technical Director for H2 Global Solutions. He has 20 years of industrial water treatment experience, both in sales and end-user roles. His focus has primarily been in the power generation industry, but has expanded into the chemical manufacturing, finished goods manufacturing and agricultural industries.
Silas Norwood
Operations Manager at H2 Global Solutions
Silas Norwood is the Operations Manager at H2 Global Solutions.