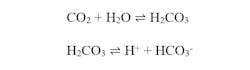The importance of industrial water and steam treatment, Part 2
Part 1 of this series emphasized the importance of makeup water treatment for the many industrial steam generators around the globe. But even with reliable makeup systems, a factor that sets most industrial steam systems apart from utility units is the increased potential for impurity ingress from condensate return.
Chemical in-leakage from reaction vessels, heat exchangers, distillation towers, evaporators and other equipment can introduce a variety of impurities to the condensate, ranging from scale-forming compounds to organics to corrosion particulates generated within the condensate return system. In this installment, we will review some of these contaminants, the problems they can cause and possible control technologies apart from direct boiler water treatment methods which will be addressed in Part 3 of this four-part series.
Scale formers
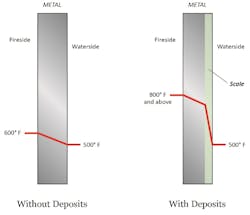
We previously observed that hardness leakage from malfunctioning makeup systems can induce scale formation in boilers. However, another source at many plants is a leaking heat exchanger that has raw water on the cooling side. In the power industry, the steam surface condenser is the primary heat exchanger where such in-leakage is possible, but industrial plants may have numerous exchangers in which cooling water could leak into the condensate. Several compounds, most notably salts of calcium and magnesium, will, at the high temperatures in boilers, form scale deposits. Figure 1 outlines in general the influence of boiler tube deposits on heat transfer.
A common result of scale formation in boiler tubes is illustrated below.
These effects become more pronounced at higher temperatures and pressures.
Ion exchange condensate polishing is a reliable technique for hardness removal, provided condensate temperatures are not too extreme. Effective for lower-pressure industrial steam unit polishing is cation exchange resin in the sodium form.1 These resins are regenerated with brine, similar to makeup water softening units.
Beware of carbon steel corrosion and corrosion product transport
Condensate systems at large industrial facilities have many miles of piping, typically of carbon steel material. Multiple compounds in the condensate can potentially cause corrosion, including carbonic acid (H2CO3) and dissolved oxygen. Carbonic acid is generated by ingress of carbon dioxide, perhaps from the boiler or from other sources in the plant. Literature references often suggest that most of the dissolved CO2 exists as hydrated molecules, but the following equations accurately represent the chemistry.
Excluding other influences, a common pH that comes from these reactions is around 5.6, but this acidity is sufficient to produce the attack shown in Figure 3.
The damage is quite visible in the Figure 3. Another serious issue in this regard is transport of corrosion products to the boiler. Typically, 90 percent or greater of carbon steel corrosion products exist as particulates. Upon reaching the boiler, these particles will deposit, typically on the hot side of the boiler tubes. Boiler water can penetrate the deposits and, upon reaching the tube metal, boil off leaving concentrated corrosive species behind. We will examine this issue, and treatment methods, in more detail in Part 4 of this series.
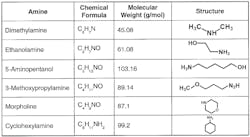
A well-known technique to neutralize carbonic acid is injection of an alkalizing amine or amines at strategic points within the condensate return network. Figure 4 shows the most common alkalizing amines.
Careful evaluation of steam-generating and condensate return system design and operation is necessary to select the most appropriate amine or amine blend. One important consideration is the presence of copper alloys in the network. Such alloys have been a common choice for heat exchanger tubes due to excellent heat transfer properties, but the presence of ammonia and oxygen can cause serious copper corrosion. Other considerations center on the efficacy of some of the compounds. For example, once popular morpholine has lost favor because the relatively high molecular weight and weaker basicity (versus the other amines) influence cost effectiveness. Amine decomposition at high temperature is another factor that can influence selection, which we will explore later in this series.
As was outlined in a previous Water Technology article, iron monitoring can be a valuable tool for evaluating the efficacy of chemical treatment programs.2 Enhanced analytical techniques now allow for the calculation of both dissolved iron and particulate iron oxides at very low concentrations utilizing a digestion method with analyses by spectrophotometry.
An advantage of this technique is the low cost of the instrumentation. However, testing does require some dedicated time by personnel trained in the procedure. On-line monitoring via nephelometry is another possibility for those wanting continuous data.2
As steam generation technology evolved in the last century for power production and industrial processes, the influence of oxygen on corrosion of boiler feedwater piping, heat exchanger tubes and other components quickly became evident. Frequently, oxygen attack is localized, where deep pits form at the site of active corrosion. This type of corrosion is very damaging and can cause equipment failure. We will examine these issues more closely in Part 3 of this series.
Condensate return organic impurities
A number of years ago, the author and a colleague were invited to an organic chemicals plant that had four 550-psig package boilers with superheaters. The steam provided energy to multiple plant heat exchangers, with recovery of most of the condensate. Each of the boiler superheaters failed, on average, every 1.5–2 years from internal deposition within, and subsequent overheating, of the tubes.
Inspection of an extracted superheater tube bundle in a laydown area revealed deposits of approximately ⅛–¼ inches in depth. Additional inspection revealed foam issuing from the saturated steam sample line of each boiler. Among the noticeable data from water/steam analyses performed by an outside vendor were total organic carbon (TOC) levels of up to 200 ppm in the condensate return. The American Society of Mechanical Engineers industrial boiler water guidelines recommend an upper limit of 0.5 ppm TOC in the feedwater of boilers at that pressure.3
No treatment processes or condensate polishing systems were in place to remove these organics (five phenol derivatives) upstream of the boilers. Based on the TOC data alone, it was easily understandable why foam was issuing from the steam sample lines, and why the superheaters rapidly accumulated deposits and failed from overheating. Furthermore, the operators had only the most rudimentary of grab sampling equipment, and could only directly analyze one of the organic derivatives. Thus, they could not readily trace what process line or lines were contaminated, and from there narrow down the problem to a specific heat exchanger or exchangers.
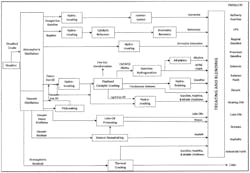
Taking the concept of condensate organics ingress to an even higher level, boilers are extensively used in refineries to supply steam to heat exchangers, steam turbine driven machinery (compressors, pumps, etc.) and offsite consumers. Figure 6 is a general schematic of the major unit operations at a refinery.4
Per the wide variety of raw, intermediate and final products, the possibilities for contaminant leakage into condensate are substantial. Obviously, different condensate return streams can contain different contaminants. Streams with hydrogen sulfide (H2S) or other highly corrosive or vile compounds are often separated and sent to the wastewater treatment plant. This scenario, of course, increases the size and complexity of the wastewater plant, and it requires a larger makeup water system to replace the dumped condensate.
For other streams containing relatively benign hydrocarbons, activated carbon filtration (ACF) may be possible for impurity removal. The huge surface area of finely ground carbon can grab many materials, and particularly non- or weakly-polar organic compounds that have a small affinity for water.5 However, other factors must be considered including condensate flow rates, condensate temperature and the kinetics of the organics-activated carbon reaction. Upfront testing is quite important to ascertain if ACF can do the job. Stripping towers are another possibility for removal of volatile organic compounds (VOCs), but then the exhaust gas may need treatment such as flaring or combustion in a furnace.
Conclusion
This article touched on just some of the potential impurities that plant personnel may have to deal with in condensate return streams. A key takeaway is that if the contaminants are allowed to enter the boiler(s), serious scaling and/or corrosion may occur. Also, the impurities may carry over with steam and cause problems in heat exchangers and turbines. Another issue that can influence condensate quality and steam heating efficiency is steam trap operation. Large steam systems may contain hundreds of steam traps, whose maintenance is vital to operation.6
References
1. Buecker, B., Koom-Dadzie, A., Barbot, E., and F. Murphy, “Makeup Water Treatment and Condensate Return: Major Influences on Chemistry Control in Co-Gen and Industrial Steam Generators”; presented at 41st Annual Electric Utility Chemistry Workshop, June 6-8, 2023, Champaign, Illinois.
2. Buecker, B., Kuruc, K., and F. Murphy, “Iron Monitoring in Industrial Steam Generating Systems”; Water Technology, Jan/Feb 2021.
3. Consensus on Operating Practices for the Control of Feedwater and Boiler Water Chemistry in Industrial and Institutional Boilers; © 2021, The American Society of Mechanical Engineers, 2 Park Avenue, New York, NY 10016, USA (www.asme.org)
4. “Detailed Study of the Petroleum Refining Category – 2019 Report”; EPA 821-R-19-008, U.S. Environmental Protection Agency, Washington, D.C., September 2019.
5. T.H. Melzer, High-Purity Water Preparation for the Semiconductor, Pharmaceutical, and Power Industries, Tall Oaks Publishing, 1997.
6. J. LaPree, “Digital Tools Help Optimize Steam Systems”; Chemical Engineering, February 2023.
[BIO]
Brad Buecker is president of Buecker & Associates, LLC, and specializes in consulting and technical writing/marketing. Most recently he served as senior technical publicist with ChemTreat, Inc. He has over four decades of experience in or supporting the power industry, much of it in steam generation chemistry, water treatment, air quality control and results engineering positions. Buecker has a B.S. in chemistry from Iowa State University with additional course work in fluid mechanics, energy and materials balances, and advanced inorganic chemistry. He has authored or co-authored over 250 articles for various technical trade magazines, and has written three books on power plant chemistry and air pollution control. He serves on the Editorial Advisory Board of Water Technology. He may be reached at [email protected].
About the Author

Brad Buecker
Brad Buecker currently serves as Senior Technical Consultant with SAMCO Technologies. He is also the owner of Buecker & Associates, LLC, which provides independent technical writing/marketing services. Buecker has many years of experience in or supporting the power industry, much of it in steam generation chemistry, water treatment, air quality control and results engineering positions with City Water, Light & Power (Springfield, Illinois) and Kansas City Power & Light Company's (now Evergy) La Cygne, Kansas, station. Additionally, his background includes 11 years with two engineering firms, Burns & McDonnell and Kiewit, and he spent two years as acting water/wastewater supervisor at a chemical plant. Buecker has a B.S. in chemistry from Iowa State University with additional course work in fluid mechanics, energy and materials balances, and advanced inorganic chemistry. He has authored or co-authored over 300 articles for various technical trade magazines, and he has written three books on power plant chemistry and air pollution control. He is a member of the ACS, AIChE, AIST, ASME, AWT, CTI, and he is active with Power-Gen International, the Electric Utility & Cogeneration Chemistry Workshop, and the International Water Conference. He can be reached at [email protected] and [email protected].