The importance of industrial water and steam treatment, Part 4
In Part 3 of this series, we examined several important issues for water/steam chemistry control in high-pressure conventional steam generators. But during the last two decades, as coal-fired power plants have been phased out and renewable generation expands, simple cycle — and especially combined cycle — power generation have provided a bridge for reliable power production and grid stability.
Usually, about two-thirds of the total power from a combined cycle plant is generated by the combustion turbine (CT), which operates on the Brayton thermodynamic cycle. The common CT fuel is natural gas, although designs are emerging to use hydrogen as a blended fuel. Capturing the exhaust heat to generate steam in a Rankine thermodynamic cycle provides the other third of generating capacity and improves plant efficiency. Modern combined cycle units may reach or even exceed 60% net efficiency, which is much better than any conventional steam-based power unit. Heat recovery steam generators (HRSGs) are also common at co-generation facilities for recovery of what otherwise might be waste heat from various processes.
While many aspects of HRSG water/steam chemistry and treatment are similar to those of conventional units, other issues influence treatment guidelines and methods. A primary factor is that most HRSGs are of a multi-pressure design, with different chemistry requirements for each circuit. We will examine some of these aspects in the following sections. A key point of discussion is the influence of flow-accelerated corrosion (FAC) on chemistry requirements.
HRSG design
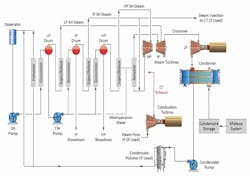
Figure 2 outlines the basic flow schematic of the most common utility HRSG design, that being the feed-forward low-pressure (FFLP) style.
Several items are worth noting up front. First, observe that the design, as is typical with HRSGs, has no feedwater heaters, apart from the economizers, in the condensate/feedwater network, unlike large fossil plants. Thus, the feedwater system is usually of all steel metallurgy. This is very important with regard to FAC control, as will be outlined. Second, in the FFLP design the low-pressure (LP) circuit primarily serves as a pre-heater for the intermediate-pressure (IP) and high-pressure (HP) evaporators. The LP evaporator produces only a small amount of steam. Because steam attemperation water is taken from the feedwater (FW) pump discharge, solid alkalis such as tri-sodium phosphate (TSP) or caustic cannot be utilized for LP evaporator pH control, as the compounds could directly enter the steam system via the attemperation sprays. Third, the load cycling duty of many combined cycle plants complicates chemistry control.
Feedwater chemistry and FAC control
During the heyday of large fossil unit evolution in the middle of the last century, the inclusion of regenerative feedwater heaters represented a significant thermodynamic improvement. The heaters recover some energy that would otherwise be lost in the condenser. Copper alloys were a common choice for heater tube material, due to copper’s excellent heat transfer properties. Feedwater networks with both carbon steel piping and copper alloys are known as mixed-metallurgy systems. In de-oxygenated water, carbon steel forms a magnetite layer (Fe3O4), while copper develops a cuprous oxide (Cu2O) layer. The cuprous oxide layer is most stable at a pH in the mid-8s, while a mid-to-upper 9 range is better for magnetite stability. Accordingly, for mixed metallurgy systems, a pH range of 8.8-9.1 became a standard guideline as a balance for controlling general corrosion between the two metals. (This range has since been adjusted to 9.1-9.3.2)
Ammonia became the common chemical for feedwater pH adjustment.
NH3 + H2O ⇌ NH4+ + OH-
In some cases, a neutralizing amine (the revised term is alkalizing amine) served as the pH conditioning chemical. Alkalizing amines offer both benefits and drawbacks, and must be carefully evaluated.3
As power boilers grew in size and complexity in the last century, researchers became certain that even trace amounts of dissolved oxygen (D.O.) during operation would cause serious metal corrosion, which is true for copper alloys in ammoniated water. Virtually all units were equipped with a mechanical deaerator. The common deaerator effluent guarantee is 7 ppb D.O. Even 7 ppb was considered excessive, however, so chemical oxygen scavenging became standard as a method to reduce D.O. to near zero. Originally, hydrazine was the oxygen scavenger/reducing agent of choice, but health concerns from handling the chemical led to its replacement with such compounds as carbohydrazide, diethylhydroxylamine (DEHA) and others.
The combination of ammonia or an amine for pH control and oxygen scavenger feed is designated as all-volatile treatment reducing (AVT(R)). The reducing chemistry maintains the magnetite layer on carbon steel and the cuprous oxide layer on copper alloys. This comfortability with AVT(R) was shattered in 1986, for “on December 9 [of that year], an elbow in the condensate system ruptured at the Surry Nuclear Power Station. The failure caused four fatalities and tens of millions of dollars in repair costs and lost revenues. FAC was found to be the cause of the failure.”4
This phenomenon is known as single-phase FAC because only water is present. (Two-phase FAC, which involves water/steam mixtures and affects deaerators, feedwater heater drains and low-pressure HRSG evaporators, is discussed in greater detail in Reference 4.) Additional single-phase FAC-induced failures over the last three decades have caused further fatalities and much damage at several power plants. FAC has been observed in many HRSGs around the globe. Gradual metal loss occurs at FAC locations, until the affected area can no longer resist the fluid pressure.
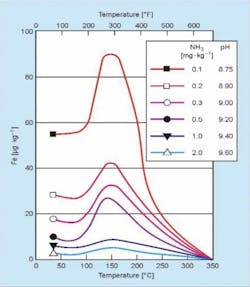
As the name flow-accelerated corrosion suggests, the attack occurs at areas of flow changes induced by piping geometry such as elbows, reducing fittings, orifices and so forth. The reducing environment produced by oxygen scavenger feed is a strong contributor, as are pH and temperature, whose effects are clearly indicated in the following famous diagram that has appeared in many technical publications (Figure 4).
The temperature influence is why FAC is so pronounced in the feedwater system and economizers of conventional units and HRSGs.
Accordingly, for high-pressure steam units without copper alloys in the feedwater system, which includes virtually all HRSGs, alternative programs to AVT(R) are now highly recommended. However, this message still has not reached many plant operators and owners around the globe.
Major FW chemistry revisions
In the late 1960s and early 1970s, chemists at supercritical units in Europe discovered that with high-purity makeup water (conductivity after cation exchange (CACE) <0.15 mS/cm), direct injection of oxygen induced formation of ferric oxide hydrate (FeOOH) on carbon steel surfaces. This red-colored oxide layer is denser and stronger than magnetite. After some evolution, this chemistry regime became known as oxygenated treatment (OT). Current guidelines from the International Association of the Properties of Water and Steam (IAPWS) call for a feedwater D.O. range of 30-150 ppb.2 OT is the feedwater treatment program of choice at most supercritical units around the world that have all-ferrous feedwater systems.
While OT can be employed for feedwater treatment in drum units including HRSGs, personnel from the Electric Power Research Institute (EPRI) developed all-volatile treatment oxidizing (AVT(O)) for high-pressure drum boiler feedwater. The primary source for oxygen is the small amount (usually) of oxygen that enters the condenser through air leaks at condenser shell penetrations, turbine/condenser expansion joints, etc. Original AVT(O) guidelines recommended <20 ppb D.O. in the condensate with a 5-10 ppb residual at the economizer. EPRI has since expanded the latter range to 5-30 ppb.5 The key aspect is that with OT or AVT(O), all surfaces in the feedwater system and economizer should have the noticeable red color mentioned above. Patches of gray-black magnetite or orange coloration indicate insufficient protection. For feed forward low-pressure HRSGs, direct oxygen injection (similar to OT applications but with lower feed rates) may be needed at the boiler feed pump to protect intermediate- and high-pressure economizer circuits from FAC.4 This need occurs because most of the dissolved oxygen in the LP inlet feedwater will flash off with the small amount of steam that is extracted.
A key point to remember is that high-purity feedwater is a requirement for AVT(O) or OT chemistry, as otherwise severe oxygen corrosion will occur. Thus, for many industrial steam generators, AVT(R) is the only choice, as impurities may enter the feedwater via the condensate return from process heat exchangers or lesser-grade makeup water systems such as sodium softeners. Feedwater and boiler water chemistry guidelines for industrial units may be found in the recently revised guidelines from the American Society of Mechanical Engineers (ASME).7 Even though industrial and institutional steam generators often operate at much lower pressure than utility units, FAC is still of potential concern.8,9
Notes about HRSG boiler water treatment
While the HRSG schematic shown in Figure 2 illustrates the most common design for combined cycle power units, a variety of configurations exist, including dual- and single-pressure drum units and once-through designs. Boiler water treatment is variable depending on these designs. We noted in Part 3 of this series that for conventional high-pressure drum boilers, tri-sodium phosphate (Na3PO4) or sometimes caustic (NaOH) are common choices for boiler water treatment. These compounds are also acceptable for pH conditioning in HRSG evaporators, but as noted earlier for the HRSG design in Figure 2, solid alkalis cannot be fed to the LP circuit per the potential direct transfer of the compounds to the steam system via the attemperator sprays. (We will examine steam chemistry in greater depth in Part 5 of this series, although for those interested in more details before then, references 10 and 11 are good sources.)
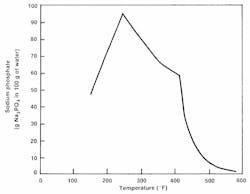
While tri-sodium phosphate is still a common choice for intermediate- and high-pressure evaporator pH conditioning, the frequent load cycling of many combined cycle units can significantly influence HRSG chemistry control. Notable is phosphate solubility, which is affected by temperature.
At high loads, much of the phosphate may precipitate (hide out) and then re-dissolve as load is reduced. Investment in comprehensive online monitoring systems and personnel who understand the data can pay off quite handsomely for monitoring this chemistry and other influences.12 The author has directly observed and heard many reports of industrial plants where the focus is almost entirely on process chemistry and engineering to the neglect of water/steam systems. Yet, boiler failures or other issues can cripple production and, much worse, perhaps jeopardize employee safety.
For lower-pressure industrial boilers or specialty waste heat steam generators, additional chemical treatments that utilize polymers to sequester, disperse and modify scale-forming compounds may be quite valuable.13,14 For example, some compounds are good at dispersing iron oxide corrosion products that enter boilers via condensate return. Dispersed particulates depart in the boiler blowdown. Other compounds inhibit scale formation that might be produced from hardness ingress. Of course, the best solution is reliable design and operation of makeup water treatment systems and condensate polishers to minimize impurity incursions.
Conclusion
In this era of expanding renewable energy sources and retirement of coal-fired power plants, combined cycle power generation and co-generation are important methods to generate the power needed for grid stability and to enhance efficiency at industrial plants. But the numerous HRSG designs and issues with load swings can make chemistry control very challenging. Minimizing flow-accelerated corrosion is a critical item for any application.
References
- B. Buecker (Tech. Ed.), “Water Essentials Handbook”; 2023. ChemTreat, Inc., Glen Allen, VA. Currently being released in digital format at www.chemtreat.com.
- International Association for the Properties of Water and Steam, Technical Guidance Document: Volatile treatments for the steam-water circuits of fossil and combined cycle/HRSG power plants (2015). Available from http://www.iapws.org.
- Shulder, S., and B. Buecker, “Remember the 3Ds of Alkalizing Amines: Dissociation, Distribution, and Decomposition”; PPCHEM JOURNAL, 2023/01.
- Guidelines for Control of Flow-Accelerated Corrosion in Fossil and Combined Cycle Power Plants, EPRI Technical Report 3002011569, the Electric Power Research Institute, Palo Alto, California, 2017. This document is available to the industry as a free report because FAC is a critically important safety issue.
- Buecker, B., and S. Shulder, “Combined Cycle and Co-Generation Water/Steam Chemistry Control”; pre-workshop seminar for the 40th Annual Electric Utility Chemistry Workshop, June 6-8, 2022, Champaign, Illinois.
- P. Sturla, Proceedings of the Fifth National Feedwater Conference, Prague, Czechoslovakia, 1973.
- Consensus on Operating Practices for the Control of Feedwater and Boiler Water Chemistry in Industrial and Institutional Boilers, 2021, The American Society of Mechanical Engineers, New York, NY.
- Buecker, B. and F. Murphy, “Breakdown: Is Flow-Accelerated Corrosion a Concern in Co-Generation Steam Generators”; Power Engineering, October 2020.
- Buecker, B., Murphy, F., and K. Kuruc, “Iron Monitoring in Industrial Steam Generating Systems”; Water Technology, Jan/Feb 2021.
- International Association for the Properties of Water and Steam, Technical Guidance Document: Phosphate and NaOH treatments for the steam-water circuits of drum boilers of fossil and combined cycle/HRSG power plants (2015).
- International Association for the Properties of Water and Steam, Technical Guidance Document: Steam Purity for Turbine Operation (2013).
- International Association for the Properties of Water and Steam, Technical Guidance Document: Instrumentation for monitoring and control of cycle chemistry for the steam-water circuits of fossil-fired and combined cycle power plants (2015).
- Buecker, B., and K. Kraetsch, “Advanced methods for controlling boiler tube corrosion and fouling – Part 1”; Hydrocarbon Processing, October 2021.
- Buecker, B., and K. Kraetsch, “Advanced methods for controlling boiler tube corrosion and fouling – Part 2”; Hydrocarbon Processing, November 2021.
Brad Buecker is president of Buecker & Associates, LLC, consulting and technical writing/marketing. Most recently he served as Senior Technical Publicist with ChemTreat, Inc. He has over four decades of experience in or supporting the power industry, much of it in steam generation chemistry, water treatment, air quality control and results engineering positions.
Buecker & Associates, LLC
About the Author

Brad Buecker
Brad Buecker currently serves as Senior Technical Consultant with SAMCO Technologies. He is also the owner of Buecker & Associates, LLC, which provides independent technical writing/marketing services. Buecker has many years of experience in or supporting the power industry, much of it in steam generation chemistry, water treatment, air quality control and results engineering positions with City Water, Light & Power (Springfield, Illinois) and Kansas City Power & Light Company's (now Evergy) La Cygne, Kansas, station. Additionally, his background includes 11 years with two engineering firms, Burns & McDonnell and Kiewit, and he spent two years as acting water/wastewater supervisor at a chemical plant. Buecker has a B.S. in chemistry from Iowa State University with additional course work in fluid mechanics, energy and materials balances, and advanced inorganic chemistry. He has authored or co-authored over 300 articles for various technical trade magazines, and he has written three books on power plant chemistry and air pollution control. He is a member of the ACS, AIChE, AIST, ASME, AWT, CTI, and he is active with Power-Gen International, the Electric Utility & Cogeneration Chemistry Workshop, and the International Water Conference. He can be reached at [email protected] and [email protected].