Minimizing Deionizer Regenerant Waste
Part 1 -- Is Zero Liquid Discharge in Sight?
by C.F. "Chubb" Michaud
Process key to water reuse, waste reduction
Ion exchange is a modern science. Until development of the strong base anion (SBA) resin in the mid-1940s, the only effective means of producing high purity water was via the millenniums old technique of distillation, a very useful process used for the recovery of salt, separation of liquids and production of pure water. It was the “origin” of the chemical industry. Distillation is also the process used by Mother Nature to supply the world's needs for potable water – the hydrologic cycle.
Modern methods of producing high purity water invariably include ion exchange. Demineralization (removal of dissolved minerals) can be done through distillation, reverse osmosis (RO), nanofiltration and/or electrodeionization (EDI) but the final deionization (DI = removal of ionic materials) is ultimately the realm of ion exchange.
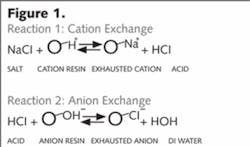
Ion exchange employs the use of charged plastic beads: a cation exchanger (negatively charged resin) that attracts and exchanges positively charged ions (cations) in exchange for hydrogen ion (H+) and an anion exchanger (positively charged resin) that attracts and exchanges negatively charged ions (anions) for a hydroxyl ion (OH-).
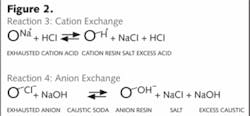
When exhausted, the resins can be restored by regenerating with acid and caustic, respectively.
Chemicals are introduced as dilute solutions of 4-6%, slowly passed through the resin beds and then the resins are rinsed to quality. A typical two bed (cation/anion) system will use 65-100 gallons of water per cubic foot of resin during the regeneration process. The focus of this article is to discuss methods of reducing that waste volume
Cutting Waste Saves Money
Years ago, the need for a zero industrial discharge stream was non-existent. Chemicals and waste discharge were done on a grand scale with little restriction. Now, people and manufacturers share the same neighborhoods, air, water and sewage treatment. They're slowly learning to live together.
Minimizing industrial waste is more “eco-friendly.” It's “green.” It's neighborly. It's also economical. Let's look at the regeneration steps for a typical two bed deionizer and consider water volumes. The first step is backwash of the cation to remove dirt and debris, as well as broken resin beads, and reclassify the bed to maintain low pressure drop. As an example, let's assume we have a two bed SAC/SBA (Type I) designed for 30 gpm – SAC for strong acid cation. We have a 24" diameter cation and a 30" anion vessel with 10 ft³ and 15 ft³ of resin, respectively.
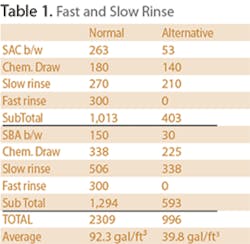
Backwash on the SAC is carried out at 5.5 gpm/ft², so we have 17.5 gpm for 15 minutes = 263 gal. Then, we start the chemical draw. If we use 4-5% HCL (assume 9 lbs/ft³ of 100% HCl), we'll use approximately 18 gal of acid per ft³ = 180 gal. Slow rinse is next and is at 150% of the chemical volume or 150% times 180 gal = 270 gal. Now comes the fast rinse at 20 gpm for 15 minutes = 300 gal. Our total cation waste = 264 + 180 + 270 + 300 = 1,013 gallons or 101.3 gal/ft³.
The anion vessel will backwash at 2 gpm/ft² or 10 gpm for 15 minutes = 150 gal. Chemical draw is 4% NaOH (assume 9 lbs of 100% NaOH/ft³) which contains about 0.4 lbs NaOH/gal, our caustic draw = 9/.4 = 22.5 gal/ft³ = 337.5 gal. Our slow rinse is again 150% of chemical draw = 506 gal. Fast rinse is again at 20 gpm for 15 min = 300 gal. So our total anion waste = 150 + 228 + 506 + 300 = 1,294 gal or 86.2 gal/ft³. Our total waste for 25 ft³ = 2,307 gal or an average of 92.3 gal/ft³. That's a good number to know. As the resins age and the rinse times increase, it will go up.
Reducing Waste Volume
Much can be done to reduce waste water by simply tightening the system. Ion exchange demineralizers are rarely used directly on raw water without some form of pre-filtration such as multi-media or granular activated carbon. This reduces the need to backwash on every cycle. We'll use the same flow rates and times but we'll now backwash (b/w) only every fifth cycle… an 80% savings on cation b/w which equals about 210 gal. We'll bump our HCl concentration to 6.5%. Again, assuming 9 lbs HCl/ft³, we'll have a chemical draw of 14 gal/ft³ and a rinse of 21 gal/ft³ for a total of 350 gal. We'll skip the fast rinse for the SAC. The reasoning follows:
SBA regenerations normally use de-cationized water from a freshly regenerated cation bed. This must be kept in mind when designing systems. An extra 90 gal of feed water per cu ft (ft³) of anion must be handled by the SAC bed. In our example, that's 1,350 gallons. If we have 25 gr/gal water, that's an extra 33,750 grains of SAC capacity or about 1.33 ft³ – an increase of about 13%. Water usage goes up accordingly for the regeneration of the SAC.
To save a little water, we'll regenerate the SBA with the unrinsed cation bed. On those occasions where we b/w, the rinse will simply be neutralized by the remaining capacity of the SBA. On those occasions where we don't, the dilute acid will be totally offset by the caustic used for the anion regeneration.
The anion chemical draw can also be more concentrated but I would use no more than 6% unless you're using hot water (120°F). At 6%, we will use 15 gal/ft³ to dilute the NaOH and slow rinse at 150% or 22.5 gal. Total chemical regeneration water is 37.5 gal/ft³ = 563 gal plus only 20% of our normal b/w (30 gal) for a total so far of 593 gal. What do you want to do about the fast rinse?
The extended slow rinse removes most of the residual chemical from the regenerated resins. By now, our cation is well rinsed but the pH from our anion is still a little high. Rather than spend that 300 gal for rinsing, we'll recirculate it back to the cation. We don't have to skimp on quality rinse because we can reuse it over and over until we rinse to quality. The results are far better than what we would have gotten with the short “fast” rinse and we saved 300 gal. So, our alternative regeneration used only about 40 gal/ft³ as shown in Table 1.
We have a reduction of 52 gal/ft³ or 57%. For our 25 cu ft system regenerating once a day, that comes to 474,500 gallons per year. At $5.00/1,000 gallons for city water, that's a savings of $2,370/yr. – just on the water bill.
Another idea is to recycle the b/w water. Store it, pump it, filter it through a bag filter to catch resin fines and use it over and over. You can even add a little chlorine to keep the bugs out and clean the resin. The water savings here is an additional 83 gal/cycle or 30,000 additional gallons per year. Combined, that's over 500,000 gallons of water saved!
Conclusion
Just by tightening up the system a bit, we've demonstrated a net water use reduction of nearly 60% and saved money to boot. But we still have almost 1,000 gallons of waste water that we have to haul away. Part 2, in the next issue, will look at making that disappear as well.
About the Author: Chubb Michaud is a 30-year veteran of the industrial water industry and holds three U.S. patents on methods of minimizing regenerant waste. A past chair of the Industrial Section and Ion Exchange Committee of the Water Quality Association, he's currently technical director of Systematix Company of Buena Park, CA. Contact: www.systematixusa.com