Sweet Plan for Sour Water
Recycling Sour Water Stripper Bottoms for Cooling Towers, Boiler Feedwater
By David Kujawski
Oil refining is dependent on the use of the distillation process. However, in the course of this procedure, condensed water accumulates in the overheads of the extraction columns. While this water is essentially distilled, the soluble gases and soluble hydrocarbons remain entrained and are in equilibrium with ionic species in the water, depending on the pH.
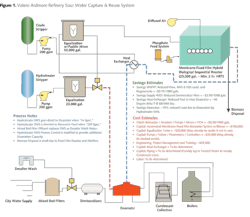
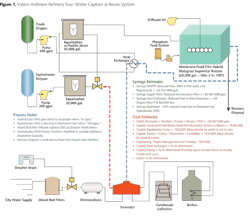
In most refineries, the overheads send water to a central collection where it is stream-stripped for bulk removal of NH3 and H2S. There are other similar small-volume sources of water that are sent to the sour water stripper (SWS) as well. In medium-sized refineries, there are typically 30 sources feeding the stripper. Some of the SWS-treated water, or "Bottoms," is sent to the desalter as wash water and from there becomes wastewater. The excess of unused SWS Bottoms for desalting is transferred directly to the wastewater treatment plant (WWTP).
With appropriate treatment of the SWS Bottoms -- to the extent where NH3, H2S, short-chain light hydrocarbons, and amines are decreased to acceptable levels -- the value of the water increases and cannot be wasted on the lower-quality needs of desalter wash water. Under normal and proper operating conditions, the SWS water has no significant levels of calcium, magnesium or iron -- the primary scale-forming inorganic contaminants of concern in boilers and cooling towers.
After specific treatment of the SWS Bottoms for these contaminants, this water is not only considered a suitable quality for steam and cooling systems but actually becomes a superior quality similar to steam condensate. Further, this captured SWS-treated water produces substantial fuel value in the form of heat. Similar to steam condensate, this water can bypass normal boiler feedwater pretreatment systems such as ion exchange or reverse osmosis (RO) and can proceed directly to the boiler deaerator.
Occasionally, SWS units are not operated or maintained correctly. A system with a significant presence of the three aforementioned inorganic cations would be overlooked as a candidate for water reuse. Most commonly, these cations enter the SWS system either by cooling water intrusion from piping and condenser leaks or by using an unsuitable water injection source in the distillation column overheads for the forcing of the dewpoint to initiate condensation.
These two conditions ultimately cause major problems in the SWS units themselves, such as the deterioration of the SWS trays, which will cause serious SWS performance problems and will require repairs. These problems almost always are short term with respect to the presence of these inorganic cations in the Bottoms, as the SWS cannot operate very long under these conditions.
In addition to boiler feedwater supply, the same considerations regarding scale formation exist as the criteria for the justification of SWS water reuse as cooling tower supply water; this also has an attractive return on investment. The validation for this cooling tower make-up can be found in the increased cycles of concentration, which would be tolerable in the cooling towers with the treated SWS water. This translates to substantial reductions in the volume of supply water used, wastewater generated and cost of chemical treatment in cooling tower operations.
The economic basis for the justification of water reuse investment at the SWS is substantially more attractive for boiler feedwater than it is for cooling tower make-up water. The return on investment for SWS Bottoms reuse as boiler feedwater is based on the reduction in the cost of treating wastewater; the decrease in the cost of supply water pretreatment; and the capture of SWS heat, which reduces deaerator heating fuel costs.
Interestingly, for those plants required to meet selenium NPDES permit limits, the routing of the SWS Bottoms to the boilers inherently extracts more than 80 to 90 percent of the total selenium load to the wastewater treatment plant, thus eliminating any selenium removal needs in the facility in almost all cases.
About the Author: David Kujawski is vice president of Refinery Water Engineering and Associates in Nederland, Texas. He has 32 years experience in water and waste treatment in over 200 industrial plants and 46 oil refineries. Kujawski holds degrees in Environmental Engineering, Chemistry, and Marketing, and over his career has held positions at Nalco, Betz-Dearborn, Baker-Petrolite, US Filter-Siemens, Chevron El Segundo Refinery, Sybron Biochemical, and Ashland Oil.