Q: What are the concerns and effects of BPA exposure from drinking water?
A: As far as I can tell, there are no reasons for concern.
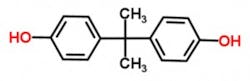
Bisphenol A (BPA) is a monomer chemical used in the production of plastic resins and in numerous other applications. Its chemical name is bis(4-hydroxyphenyl)propane, and its chemical structure is shown in Figure 1.
Figure 1. BPA’s molecular chemical structure. Graphic courtesy of chemspider.com/Chemical-Structure.6371.html (accessed on July 13)
BPA is present in many consumer products, so high potential for low-level exposure exists. Infants who drink formula from polycarbonate baby bottles generally have higher exposures per kilogram body weight than adults.
The molecular structure in Figure 1 shows that BPA is a relatively simple chemical — two phenols linked back to back through a propane unit. It is a white crystalline solid that melts at about 158˚C (318˚F). Its molecular weight is 228.29 daltons; water solubility is 120 to 300 parts per million at 21.5˚C (71˚F). It is manufactured easily by con-densing two molecules of phenol with acetone. It has many uses. The largest is in the production of polycarbonate plastics (a glass substitute) when it is reacted with phos-gene, and epoxies by reaction with epichlorohydrin.
All four of the reactants are common, inexpensive chemicals, and world production approaches 10 billion pounds per year. Epoxy and polycarbonate plastics are extremely stable products, but 100 percent reaction does not occur when the polymers are formed, so a small amount of the BPA will remain and be leachable to food and water during contact. It is biodegraded during waste treatment and by numerous environmental microorganisms.
BPA uses
Apart from plastics production, BPA has many other applications in its free state as an antioxidant for polyvinyl chloride and color developer in carbonless copy paper, and on thermal receipt paper. It is also used in the manufacture of:
- Printed circuit boards
- Electronic equipment
- Paints
- Adhesives
Human exposure to BPA
About 93 percent of tested Americans older than age six had detectable BPA or BPA metabolites in their urine. Exposure can occur by ingestion from diet, canned foods with epoxy liners and contact with products such as thermal paper with free BPA. It is efficiently absorbed through the skin. In addition, exposure can occur from extraction to liquids in polycarbonate containers, including baby bottles and water carboys.
Daily adult intake from food, beverage and environment has been estimated at about 0.4 microgram (µg) up to about 1.5 µg per kilogram (kg) of body weight per day. This is about 28 µg to 100 µg per day for a 70 kg adult based upon urine concentrations. Some occupational exposures can be up to about 100 µg/kg per day, or 7,000 µg per day. Urine measurements are usually calculated relative to the release of creatinine, which is a normal waste product that indicates kidney function and is used as a baseline standard so that variations in urine output can be normalized.
Drinking water
Public drinking water is a potential source but is a minor contributor (generally less than 1 percent) to total exposure. The U.S. Environmental Protection Agency (EPA) reported that concentrations in U.S. drinking water are typically well below 1 µg per liter (L) or 1 part per billion (ppb) when detected. Levels in treated wastewater have been reported at up to 22 ppb. Concentrations of BPA in U.S. drinking water had a median range of 0.005 to less than 0.1 ppb and an overall range of less than 0.1 to 0.42 ppb.
Surface waters ranged from 0.0009 to 12 ppb. Groundwater levels ranged from 0.006 to 2.55 ppb and wastewater from 0.0036 to 50 ppb. If BPA, a phenol, were present in chlorinated drinking water, it would likely be converted to chlorinated forms. BPA and chlorinated BPA would be readily removed from water by activated carbon or reverse osmosis membranes.
Toxicology
BPA is a hormonally active chemical and an estrogen (17β-estradiol) mimic. It does not bind strongly to the estrogen receptor. The relative estrogenic potency is generally considered to be on the order of one ten-thousandth of estrogen, and its primary glucuronide metabolite is considered non-estrogenic. Numerous dose-related health effects have been reported in cell culture and in vivo animal studies, but human effects at low environmental doses have been more elusive.
BPA has been the subject of thousands of studies over the years, and the interpretations have been controversial. These reports from mostly in vitro and animal studies cover its effects on everything including:
- Neurological
- Thyroid
- Prostate
- Breast
- Testis
- Mammary gland
- Body size
- Brain structure
- Heart disease
- Diabetes
- Oxidative stress
- The behavior of laboratory animals
Some have since been discounted by later studies. The question becomes what are the concerns for human exposure at typical and elevated levels?
Levels of concern
Several committees have reviewed the state of the science and often arrived at different conclusions. A 2006 government-sponsored committee concluded that BPA at average concentration levels in the population had caused harm in laboratory animals.
On the other hand, a 2008 report from the National Toxicology Program expressed "some" concern for infants, and generally "negligible to minimal" concerns for birth defects. The concern was "minimal" for highly exposed occupational situations.
Numerous calculations of "intake limits" have been reported. The EPA, U.S. Food and Drug Administration (FDA), and European Food Safety Agency (EFSA) values tend to be in the range of 50 µg/kg per day or higher, and they incorporate safety/uncertainty factors ranging from 100 to 1,000. The EFSA recently lowered its value to 4 µg/kg per day and concluded that it was three to five times the highest exposure estimates.
A few years ago, California’s Office of Environmental Health Hazard Assessment (OEHHA) attempted to list BPA for Proposition 65 notification, after its advisory committee declined to identify it as a hazard. OEHHA was litigated, removed it, formed a new advisory committee which voted to list it, and has recently done so. The original proposed listing was based on reproductive concerns at an acceptable value of 290 µg per day from several rodent reproductive and developmental toxicity studies with a no-observed-adverse-effect level of 5 mg/kg per day, and a 1,000 margin of safety for a 58-kg (128-pound) pregnant woman. That value is much higher than the total daily population exposure, indicating negligible risk from ambient exposures.
Metabolism
BPA is rapidly metabolized in the liver and eliminated either as the parent, or conjugated as the glucuronide or sulfate. Researchers from the National Center for Toxicological Research built mathematical models of how it is processed, metabolized and eliminated. They showed that BPA is rapidly metabolized and eliminated through feces and urine. They also did not find evidence of BPA toxicity at low doses in rodent studies, including doses at about human exposure levels.
In addition, the models could not measure BPA being transferred from pregnant rodent mothers to fetuses after being fed amounts that were 100 to 1,000 times more than people are exposed to from food. The researchers estimated that human infants were exposed from 84 to 92 percent less than previously estimated.
BPA is metabolized differently by humans and other primates than by rodents. Humans produce more of the BPA-glucuronide compared to rodents. Free BPA is weakly estrogenic whereas BPA-glucuronide is not estrogenic. The relative amounts of free BPA and BPA-glucuronide in humans and other primates versus rats indicate that rodents may be more sensitive than humans to BPA. This indication should be considered when attempting to extrapolate data from rodent studies to humans. The EPA also states that more than 90 percent of the BPA found in the urine of premature infants was in the BPA-glucuronide form, indicating that infants can metabolize BPA to this non-estrogenic form.
Regulations
Several countries, including the U.S. and Canada, have banned BPA in baby bottles, usually in the interest of caution, because of the relatively high intake to body weight ratios for infants versus adults and because alternative materials are available.
The EPA has produced an action plan for BPA primarily because it is a high-volume chemical and therefore can have substantial releases to the environment (more than 1 million pounds per year) and because alternatives may be available for some uses. Food-related packaging is less than 5 percent of the BPA used in the U.S., but it accounts for most of the human population’s exposure, except perhaps for dermal contact for some people with high occupational exposure. The EPA has deferred to the FDA on food-related issues, but it will address environmental releases and non-food packaging and whether human environmental exposure is significant, which does not seem to be the case. The EPA recently included BPA as a candidate for the next Contaminant Candidate List (CCL4).
Environmental effects
Because BPA has weak estrogenic properties, concerns about environmental effects in freshwater and marine aquatic organisms must be considered. The European Union computed a predicted no effect concentration (PNEC) of 7.5 µg/L and included an uncertainty/safety factor of 5 to arrive at a PNEC of 1.5 µg/L.
The PNECs for Canada and Japan are 0.175 µg/L and 1.6 µg/L, demonstrating that all risk assessments contain a substantial element of judgment and different margins of safety beyond the science.
Conclusion
BPA continues to be a controversial topic in the press and the scientific and regulatory community. Thousands of toxicological studies have been performed, but a universal consensus has not been reached on whether human impact exists from low-dose exposure from all sources.
The FDA has conducted extensive studies, and it has stated several times that BPA is not harmful at the current exposure levels. The EPA gives it a fairly low priority, indicating low potency compared to estrogen, but it has initiated an action plan of potential information gathering activities. It has listed BPA as a candidate for the CCL4.
However, BPA does not seem to fit the selection criteria on exposure and risk. California’s OEHHA just relisted it for Proposition 65 notification after its current reproductive toxicology advisory committee recommended it.
The good news is that, regardless of the scientific and regulatory debates, drinking water is a negligible source of BPA exposure compared to total dietary and environmental exposure. In reality the case for human risks at normal levels seems extremely weak. Never-the-less, more studies continue to be funded.
Dr. Joseph Cotruvo is president of Joseph Cotruvo and Associates, LLC, Water, Environment and Public Health Consultants. He is a former director of the U.S. EPA Drinking Water Standards Division.