Reverse osmosis system scale management boosts RO recovery
Reverse osmosis (RO) is the workhorse of desalination. Its versatility and ubiquity are advancing into more demanding applications, particularly the industrial wastewater treatment space. Industrial wastewater poses challenges for RO, most of all, scale and brine management. Scale limits recovery, leading to increased brine volumes and disposal or downstream brine concentration costs. When unchecked, scale adds operational risk for increased membrane maintenance and replacement. If water chemistry is variable — as can often be the case in industrial applications — systems are operated conservatively, generating excess brine or put at risk of damage.
This article reviews the innovations in RO processes aimed at defending against scale while boosting recovery. A series of RO processes have been released that delay or mitigate scale, but do not entirely remove this risk. The following modernized method to remove the scale risk can entirely be partnered with any RO process. The result is a viable option to economically drive RO systems to their osmotic pressure limits.
RO and UHP-RO
The reverse osmosis operating principle is straightforward: High pressure is used to drive water through specially engineered semipermeable membranes that reject salt ions. Recovery increases with higher brine concentration relative to inlet salinity, squeezing more freshwater from the salt water. Osmotic pressure also increases with brine concentration, which requires higher driving pressures and reduces freshwater permeate flux, requiring larger membrane area. Historically, there are three pressure classes for RO membranes: 300 psi, 600 psi and 1200 psi. The higher the pressure class, the higher the brine concentration potential on a nonscaling fluid. However, RO membrane vendors are innovating ultra-high-pressure reverse osmosis (UHP-RO) spiral wound membranes capable of 1800 psi (120 bar). UHP-RO enables brine concentrations up to 130,000 mg/L total dissolved solids (TDS) for NaCl and 150,000 mg/L for Na2SO4 — a 50 percent decrease in brine volume relative to previous generations. This produces cleaner water and limits downstream brine disposal or brine treatment costs.
This assumes no scaling or fouling compounds. RO brine concentration will be limited by either osmotic pressure increase with salinity, which decays permeate flux to an unsuitable level, or scaling ions or organic fouling that can block the membrane. Most industrial RO applications are scale-limited, for example, by silica, calcium sulfate, phosphate, fluoride, iron or barium salts. Organic fouling can be managed by pretreatment, a biocide program or automated high-pH washes. It is important to note that pretreatment for extremely high organics or solvents can make RO uneconomical, which may be the case in certain oil and gas produced water applications.
Ionic scale can affect end users in three ways: reliability challenges, leading to frequent cleaning or shortened membrane life; recovery left on the table generating more brine than required; or RO membrane fouling caused by indirect effects such as coagulants intended to help remove precipitated scale upstream. Problems are compounded in industrial wastewater where the feed chemistry varies, requiring constant monitoring.
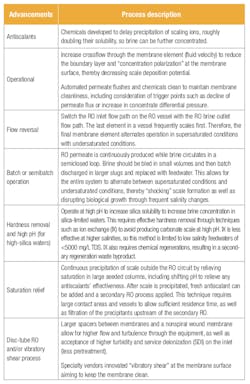
Table 1. Comparison of advanced RO processes and configurations.
Advanced innovations in reverse osmosis
Walking the challenging tightrope of scale management can be steadied with a variety of different techniques. Table 1 provides a summary of these techniques. A new technique (described in the next section) can be partnered with any of the Table 1 processes to limit risk or further boost RO recovery.
The processes shown in Table 1 all share one common thread: mitigate scale formation. However, recovery may still be below osmotic pressure limits with brine concentration potential left on the table. Operators may also still be walking a tightrope because scaling compounds are circulating in a sensitive RO system. Changes in water chemistry or incorrect operation can nudge one off the scale tightrope — a misstep that can be damaging and costly.
Figure 1. Integration of RO and advanced chemical softening systems to increase RO recovery reliably.
Chemical softening
There is a hybrid solution that removes the tightrope entirely as shown in Figure 1. First, achieve initial recovery through a primary reverse osmosis (RO-1) step, effectively reducing the volume for downstream processes. The techniques in Table 1 are applicable, but do not need to be pushed to their limit. After primary RO-1 recovery, chemically soften the brine to extract the scaling compounds before another secondary RO (RO-2) step that boosts recovery to the osmotic pressure limit of RO.
Chemical softening has been considered mature technology for over 50 years, but still has its challenges. Surprisingly, it has received little attention for innovation. A designer should be careful not to extract textbook chemical softening and expect it to be reliable with RO, largely because the coagulants and flocculants considered safe for physical-chemical processes can foul a downstream membrane system. (An article on saltworkstech.com includes a more detailed background on chemical softening including procedures, chemical addition types and pictures of settling times.)
By applying modern automation and separation processes to solve these challenges, Saltworks’ BrineRefine is a compact RO-friendly chemical softener. Innovations include:
- Reduced footprint by replacing the reaction tanks and clarifier with an inline processing system
- Precision dosing by modern automation to prevent overdosing (wasted chemicals and added sludge) or underdosing (scale risk), including the ability to adjust with changing upstream chemistry
- Elimination of coagulants and floc, which can foul downstream ROs — forming a gel on the membrane that would require frequent chemical cleans
- Replacement of manually intensive sludge management techniques with semi-automated solids management technology, borrowed from a crystallizer technology – the SaltMaker
The controls communicate with any upstream and downstream RO to optimize total system performance. Wastewater treatment plants should not be designed or controlled as a linear train of independent unit operations passing water to each other. They should be considered as an integrated system, with each unit operation adjusting to its "sweet spot" as upstream conditions change. The systems’ view enables optimization of total system economics, reliability, energy, chemical consumption, recovery and capacity.
The system does not remove soluble organics. As RO recovery is driven to new limits, users must be cautious of organic accumulation in the secondary RO loop. This can require an organic treatment "kidney" in the circuit or plans for automated clean-in-place (CIP). Engineers skilled in high recovery treatment can advise for each specific situation.
Figure 2. Significance of scale accumulation in pipe
Tips to optimize economics and achieve high recovery reliably
- Employ a primary RO to reduce volume for downstream processes. Ensure the controls of each unit operation are intelligently integrated such that downstream processes adjust to upstream changes in operations or feedwater to prevent reliability challenges or lost recovery.
- Before aiming for high recovery, first understand brine disposal/brine management costs. This will show a facility’s breakeven point between brine minimization versus brine disposal. Tools are available to calculate these costs.
- Understand the solids composition and its disposal costs. The more chemicals added, the more solid waste is generated for disposal. If radium or normally occurring radioactive material (NORMs) are present, they will coprecipitate with barium and could produce a "radioactively hot" final solid. Water can be tested to manage this risk.
- Do not remove all scaling compounds to minimal achievable levels, but remove them to where brine concentration leaves the system with a comfortable margin for maximized brine recovery and reliability, such as within 85 percent of scaling potential.
- Review site-delivered chemical costs and compatibility with water chemistry. For example, when approaching a high pH step to remove heavy metals, fluoride or silica, if there are high levels of magnesium in the wastewater, raising pH by lime would be a lower cost option. If magnesium is low, sodium hydroxide may be more cost-effective for the high pH step due to reduced subsequent soda ash costs. Skilled practitioners can easily run scenarios and calculate optimal costs.
- Avoid coagulants (aluminum and iron salts) and flocculants (polymers) where possible. They are an
additional chemical cost, require careful hydraulic shear management, and most importantly, they can put the downstream RO membranes at great risk of fouling.
Conclusion
Innovation is driving down the cost of treating industrial salt water. Membrane systems cost five to 10 times less than thermal brine concentration evaporators, so it is important to maximize recovery of any membrane system upstream of expensive evaporators or offsite disposal. This article provided techniques to safely and economically maximize membrane system brine concentration to the new TDS limits of UHP-RO membranes (130,000 to 150,000 mg/L).