Testing activated carbon performance for difficult adsorbates: Trace capacity number: Part one
Small molecules are difficult to predict adsorption
A new Trace Capacity Number Gas-phase or TCN test method is available to help determine the best activated carbon for removal of trace concentrations of water soluble small molecules and gas-phase applications. TCN drinking water applications include removing methyl-tertiarybutyl ether (MTBE), trihaolmethanes, vinyl chloride, methylene chloride, methanol and other small water soluble molecules. TCN can be used to evaluate adsorptive trace capacity of activated- or reactivated carbons. TCN is designed to complement the Gravimetric Adsorption Energy Distribution or GAED full characterization test method.1 Figure 1 shows the challenge gas as chemical structures used in the two test methods.
TCN values correlate directly with aqueous MTBE removal from drinking water, but iodine numbers do not correlate. Published test procedures to determine iodine activity numbers of activated carbons produce results that do not correlate with field studies for target small molecule removal from drinking water. Tetrafluromethane (TFM) or carbon tetrafluoride activity or TCN is the ratio (g/100 cc of activated carbon) mass of tetrafluromethane adsorbed by a volume of activated carbon sample when the carbon is saturated with tetrafluromethane under conditions of this test method. An activated carbon bed of known volume and mass is saturated with tetrafluoromethane vapor. The mass adsorbed at saturation is measured and reported as mass of tetrafluoromethane per unit volume of carbon.
Trace capacity number compared to GAED
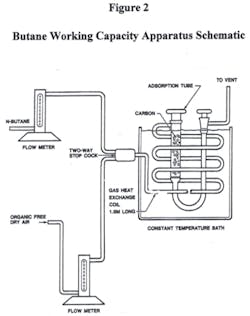
Trace capacity number or TCN test method is a logical extension of earlier test method Gravimetric Adsorption Energy Distribution or GAED full characterization of physical adsorbents test method.1 GAED determines the pore sizes of adsorption spaces and their corresponding adsorption energy distribution from the largest to the smallest adsorption spaces, but the GAED equipment is relatively complex and expensive. The terms pore and adsorption space are used interchangeably. GAED provides the characteristic curve for a material, which can be described by its polynomial equation, to yield isotherms for compounds of interest at any temperatures desired.1 Isotherms provide the most important information, it is a "GO" or "NO GO" carbon application and it provides an estimate of the carbon use rate. GAED also provides pore size distribution, BET surface area, thermogravimetric analysis and trace- and mid-capacity numbers for the tested sample. The TCN method described in this article uses relatively low cost, readily available equipment compared to GAED. TCN equipment set-up is accepted to provide ASTM International butane and carbon tetrachloride activity numbers (see Figures 2 and 3).2
TCN activity (g/100 cc) values are lower than butane activity because only the smallest adsorption spaces, which have the highest adsorption energy, are capable of physical adsorption of tetrafluoromethane at 25o C. Laboratory sample preparation techniques to increase TCN resolution have been incorporated to deal with relatively low mass TFM loadings on activated carbons. Accurate TCN activity values depend on these highest adsorption energy (HAE) pore spaces’ cleanliness: No adsorbates present, water or concentrated air in the HAE pores. Activated carbons can concentrate ambient atmosphere air about seven-fold in these highest adsorption energy pores. Water loading needs to be less than 0.1 wt percent, and these pores need to be empty of adsorbates, including concentrated air and char, when the pre-weighed sample tube (see Figure 3) is re-weighed with the sample. This necessary sample conditioning is accomplished by weighing the sample warm, while the sample is over 70o C, when filling the sample tube (Figure 3).
Granular activated carbon (GAC) TCN values are smaller than butane activity values, about five to 10 times smaller. These five to 10 times comparative values are preliminary and need more industry collaborative evaluations. Standard ASTM International butane activity runs flow butane in a bottom to top flow (see Figure 3). But, for TCN runs flowing tetrafluoromethane (TFM) needs to be reversed top to bottom, this flow uses the fritted disk or equivalent in the bottom of the sample tube to contain any carbon dust. With the relatively small TFM loading mass, not losing any GAC sample mass is important to assure accurate TCN values. It is also important to tightly pack the activated carbon sample into the sample tube; a vibrating plate is used as described in ASTM method D 2854 to accomplish proper packing.
It is important to know that popular physical adsorption specifications, iodine number and BET surface area, have little relevance to activated carbon adsorption performance for small molecule applications. Apparently this is true because most of the iodine and surface area value, mg/g and m2/g respectively, is derived from irrelevant larger pore sizes not needed for small molecule applications. The TCN test method has more relevant specificity compared to iodine and surface area values because TCN only evaluates the pore sizes needed for these difficult adsorbate applications.
This TCN method is a gas-phase test method used to predict aqueous-phase GAC performance in addition to predicting gas-phase applications. Activated carbons are used in both aqueous- and gas-phase applications. Sometimes the same activated carbon is used in water-, solvent- or gas-phase applications. Typically, particle sizes are larger for gas-phase applications compared to liquid applications to maintain high flow rates and avoid high pressure. Polanyi provided original seminal work to connect gas-phase and aqueous-phase physical adsorption phenomena.3 Polanyi concepts are embedded in the GAED test method. Polanyi, and many later scientists, intellectual work product enables today’s marketplace to use either gas- or aqueous-phase test methods to predict performance for aqueous-, solvent- or vapor-phase physical adsorption applications.
In aqueous applications it is very important to assure complete wetting of granular activated carbon or GAC porous structures. The smaller the pore, the harder it is to wet because it has more compressed air that needs to be displaced with water. The small micro-pores contain seven atmospheres of air pressure above ambient. Incomplete wetting is the most common reason for carbon not working.4 Aqueous adsorbates must diffuse from bulk water to active adsorption sites with 1- to 5- nm wall-to-wall graphitic platelet adsorption spaces, or pores. To assure complete micro-pore wetting it is recommended that GAC is soaked in water for 72 hours with several drainings and refills with water, before field use to assure complete wetting.4 Hot water wetting decreases this needed time, because hot water penetrates GAC better. You can also purchase properly wetted activated carbon instead of dry; this allows the carbon to be used immediately in the field by eliminating the wetting process. Once compressed air bubbles are formed in the bowels of carbon particles, trapped compressed air can stabilize for a long time, even years, which effectively eliminates their needed adsorption spaces and results in pre-mature adsorbate breakthrough. If a significant portion of a GAC column has trapped compressed air, the water flows around the collective air bubble and loses carbon contact. So if the GAC has a high TCN value, but field operations do not properly wet the GAC, it will not perform to its potential small molecule capability.
In part two of this article, we'll pick up the discussion with MTBE's unintended consequences.
References:
- Henry Nowicki provides public and on-site short courses two to three times a year titled, “Activated Carbon Adsorption: Principles, Practices, and Opportunities.” During this course, PACS covers specific course attendee issues to solve their problems in addition to the basic course. The GAED is covered in this course.
- ASTM International standards of activated carbon test methods: D 5742 Test Method for Determination of Butane Activity of Activated Carbon, D 5228 Test Method for Determination of Butane Working Capacity of Activated Carbon, D 3467 Test Method for Carbon Tetrachloride Activity of Activated Carbon, D 2854 Test Method for Apparent Density of Activated Carbon, D 2867 Test Methods for Moisture in Activated Carbons, D 5919 Practice for Determination of Adsorptive Capacity of Activated Carbon by a Micro-Isotherm Technique for Adsorbates at ppb Concentrations.
- M. Polanyi, Verh Deut Physik. Ges. 16 1012 (1914) and 18, 55 (1916).
- Wayne Schuliger, “Activated Carbon is not the Problem.” International Activated Carbon Conference. September 1999 and his one-day PACS public and on-site course titled: “Design, Operation, Troubleshooting Aqueous- and Vapor-phase Activated Carbon Adsorbers.”
Henry Nowicki, PhD, MBA, is PACS Laboratories president and senior scientist and has been awarded nine government SBIR contracts for R&D on new activated carbon products. PACS provides one-stop shopping for activated carbon testing, consulting and training services. Nowicki provides the introductory course for PACS Activated Carbon School titled “Activated Carbon Adsorption: Principles, Practices, and Opportunities.” He has been an expert witness for environmental, safety and activated carbon cases. He is technical chairperson for the annual "International Activated Carbon Conference and Carbon Courses." Hope to see you at next carbon conference in Pittsburgh, Pa. September 24-25, 2014. Author may be reached at (724) 457-6576 or www.pacslabs.com or e-mail [email protected].