Testing activated carbon performance for difficult adsorbates: Trace capacity number: Part two
Editor's note: This article is a continuation of an article from April 2014, which can be found on WaterTechOnline.com.
MTBE unintended consequences
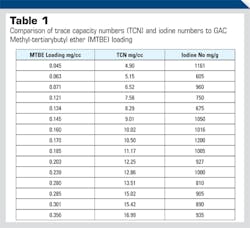
Methyl-tertiarybutyl-ether (MTBE) was added to gasoline as a fuel additive to facilitate ignition and improve air quality. MTBE has contaminated significant groundwater drinking water sources, because it is very water soluble and migrates relatively quickly from underground leaks compared to hydrocarbons, from leaking storage tanks. It has a bad taste and odor in drinking water at low trace concentrations and thus needs to be removed. GAC removes MTBE, but its performance lot-to-lot or vendor-to-vendor is not predictable using iodine or butane numbers. Iodine numbers do not predict MTBE loading capacity for GAC supplies, but Trace Capacity Numbers do correlate (see Table 1). Iodine and butane ASTM tests fill a wide spectrum of adsorption spaces, but tetrafluoromethane is only taken up by small, high adsorption energy adsorption spaces. Sites needed for MTBE adsorption. Thus, iodine is best used as a performance predictor when the real-world application fills up the total pore volume and TCN is more appropriate for specific applications that need selectively only small HAE adsorption spaces.
Comparison of TCN and iodine numbers
Trace levels of aqueous MTBE adsorption are unpredictable for commercial activated carbons based on iodine numbers, because MTBE is very water soluble and present at low ppb concentrations. The more water soluble a potential GAC adsorbate is the more difficult it is to adsorb, and its loading capacity is lower. The closer the adsorbate is to saturated concentration, the higher the GAC loading, above saturation you get two phases of liquids. GAC has a low and variable MTBE removal capacity when compared to iodine, but loading capacity is more predictable when TCN is used as the correlation with MTBE removals from water. Results of this comparative evaluation are shown in Table 1 below for several activated carbon samples. This data set tells us that TCN values go up with MTBE increased loading or capacity and the iodine number does not correlate with MTBE loading.
The above data table shows that different activated carbon samples TCN correlates directly with MTBE removal from clean water. It also clearly shows that the iodine number has no correlation with MTBE removal performance.
This trace capacity number test method is expected to be most useful for drinking water from groundwater supply sources. Surface water sources tend to have more organic contaminants and thus we expect competition for the limited number of high adsorption energy sites. HAE sites are required for physical adsorption of small water soluble organic compounds, like MTBE, THM, vinyl chloride and other small water soluble compounds. Since about half of the U.S. drinking water sources are groundwater supplies, TCN test method should be useful.
Discussion
Accurate test methods to predict activated carbon’s specific application performance are critical for activated carbon users and manufacturers to select the best carbon. Activated carbon has an amazing long list of applications. All carbons are not the same, you need to choose wisely. A carbon which lasts two to three times longer than a competitor carbon in a given application is more useful than a carbon which breaks through earlier. Early GAC breakthrough results in repurchase of unused carbon and change out of the spent used carbon and its disposal. The MTBE used GAC more likely than not still has a good iodine number, when the TCN has been consumed and the MTBE filled micropores are not available to provide more needed MTBE physical adsorption. Obviously, MTBE relatively lightly used carbon can be reactivated for many uses or the municipality could use this MTBE used carbon at their municipal wastewater plant. Presently municipal wastewater plants do not provide significant tertiary treatment with activated carbon, yet their effluents are well known to have deleterious organic compounds that could be removed very well with activated carbon.1
It is generally accepted that adsorption space or pores need to be 1.3 to 1.8 times larger than the adsorbing molecules volume. Thus, small molecules are best adsorbed in the smallest high adsorption energy pores. The term pore and adsorption space are used interchangeably. Physical adsorption of small molecules is a nanotechnology phenomenon. Adsorption of molecules depends on a power of three molecular distance between adsorbate and graphitic platelet, i.e. one-molecular layer away is a relative adsorption force of one, two-molecular layers the relative force is 1/8, three-molecular layers away from the GAC graphitic platelet the relative adsorption force compared to one-molecular layer is one twenty-seventh (1/27th) and three molecular layers away from graphitic platelet results in negligible physical adsorption.1 The mass ratio of adsorbate to solid becomes of significant magnification when a significant fraction of the atoms of the solid is at a surface.
Physical adsorption of solvents out of water is enhanced liquefaction, that is, the adsorbate comes out of the water in a liquid-like state in micro-pores well before it is concentrated to saturation in micro-pores. Adsorbates are concentrated in specific adsorption spaces by London force fields from graphitic platelets.5 Polarizability of adsorbates determine how well they come out of water as liquid or solids, like the physical state of the molecules in bottles in the chemical store room. Polarizability is a direct reflection of refractive index.
Conclusion
If you are having problems getting expected GAC performance (gallons treated per pound or volume of activated carbon) removing small water soluble target molecules using iodine numbers as the predictor to evaluate incoming GAC, you need to consider TCN test method to help solve this problem. Even if you are not having this problem, you need to know about TCN and GAED advanced testing methods and what they can do for your future physical adsorption process applications. TCN uses common industry standard equipment used for ASTM International butane and carbon tetrachloride activity tests. GAED full characterization also enables determination of TCN values, but uses more complex equipment and software.
References:
1. Dr. Henry Nowicki provides public and on-site short courses two to three times a year titled, “Activated Carbon Adsorption: Principles, Practices, and Opportunities.” During this course, we cover specific course attendee issues to solve their problems in addition to the basic course. The GAED is covered in this course.
2. ASTM International standards of activated carbon test methods: D 5742 Test Method for Determination of Butane Activity of Activated Carbon, D 5228 Test Method for Determination of Butane Working Capacity of Activated Carbon, D 3467 Test Method for Carbon Tetrachloride Activity of Activated Carbon, D 2854 Test Method for Apparent Density of Activated Carbon, D 2867 Test Methods for Moisture in Activated Carbons, D 5919 Practice for Determination of Adsorptive Capacity of Activated Carbon by a Micro-Isotherm Technique for Adsorbates at ppb Concentrations.
3. M. Polanyi, Verh Deut Physik. Ges. 16 1012 (1914) and 18, 55 (1916).
4. Wayne Schuliger, “Activated Carbon is not the Problem.” International Activated Carbon Conference. September 1999 and his one-day PACS public and on-site course titled, “Design, Operation, Troubleshooting Aqueous- and Vapor-phase Activated Carbon Adsorbers.”
5. F. London, Z. Phys. Chem. B11, 222 (1930).
Henry Nowicki, Ph.D., MBA, is PACS Laboratories' president and senior scientist and has been awarded nine government SBIR contracts for R&D on new activated carbon products. PACS provides one-stop shopping for activated carbon testing, consulting and training services. Nowicki provides the introductory course for PACS Activated Carbon School titled, “Activated Carbon Adsorption: Principles, Practices, and Opportunities.” He has been an expert witness for environmental, safety and activated carbon cases. He is technical chairperson for the annual International Activated Carbon Conference and Carbon Courses. Author may be reached at (724) 457-6576 or www.pacslabs.com or e-mail [email protected].