Strong base anion resins (SBA), operated in the chloride form, can remove many contaminants from drinking water. These contaminants include arsenic V (also known as arsenate), nitrate, chromate, fluoride and uranium. The two types of strong base anion exchange resins commonly used today are Type 1 and Type 2 strongly basic anion exchange resins. Both resins can be used to remove the contaminants listed above.
The Type 1 resin derives its ion exchange capabilities from the trimethylamine (TMA) functional group. The Type 2 resin derives its functionality from the dimethylethanolamine (DMEA) group. The TMA in Type 1 anion resins has the potential to contribute fishy odor to the treated water under alkaline conditions. The Type 2 anion resins do not have the potential to contribute odors to the water and because of this are preferred in many drinking water applications.
The relative order of affinity of these strong base anion resins for some common ions in drinking water is: Uranium/Perchlorate> Sulfate/Chromium > Arsenate> Nitrate> Chloride> Bicarbonate> Fluoride.
Looking at these affinity relationships, you can see why the standard anion resins are limited in most applications by the amount of sulfate in the water. Sulfate is the strongest held bulk anion and is almost always present in much higher amounts than the ions that we are trying to remove in drinking water treatment: Fluoride, nitrate, arsenic, selenium, chromium, etc. Aside from nitrate, the other contaminants listed are considered trace contaminants and if present in the water, are reported in microgram per liter (µg/l) or part per billion (ppb) levels. The bulk ions, sulfate, nitrate (if any), chloride and alkalinity are reported in milligrams per liter (mg/l) or part per million (ppm).
The exchangeable ion on the strong base anion is the chloride ion. The chlorides initially replace all anions, so the chloride level in the effluent of the unit is equal to the Total Dissolved Solids (TDS). Bicarbonates will be exchanged for chlorides in the first part of the run, and then pushed off the resin in the latter part of the run. The pH will be reduced during the first part of the run due to the removal of alkalinity and increased above the influent pH once bicarbonates (alkalinity) begin to leak. Chlorides will be equal to the sum of sulfate, chloride, alkalinity and nitrate during the first part of run and equal to sulfate, chloride and nitrate during the latter part of the run.
The resin is typically regenerated with dilute brine, about five to 10 percent strength, usually at 10 to 15 pounds per cubic foot. Product water can be buffered by addition of soda ash (Na2CO3) to the brine tank (one pound of soda ash to nine pounds of salt). When operated in the chloride cycle (sodium chloride regeneration), the resin will not exchange for carbon dioxide (CO2) or silica (SiO2). Recommended service flow rate is three to five gallons per minute per cubic foot (gpm/ft3.)
What if we didn’t make adjustments for the pH depression which is observed during the initial portion of the service cycle when the alkalinity is being removed?
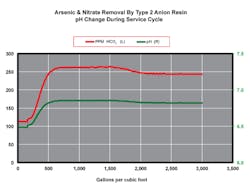
Figure 1 illustrates the ionic breakthrough profile from the effluent of a strong base anion unit. The graph is run out past exhaustion so that the chromatographic dumping of each ion can be observed. Looking closely at the beginning of the run, we see that the alkalinity (HCO3) is the first ion to start appearing in significant levels in the effluent other than chloride. It is during this period that the pH is lowered.
Figure 2 shows the alkalinity and pH relationships in the breakthrough profile of the effluent. The pH is as low as 6.5 at the start of the run before it climbs to 6.7 later in the run. In some waters, the pH drop can be an entire pH unit or more. How can we use this to our advantage?
Arsenic selective medias
The selective arsenic medias available today are based on metal oxide chemistry, usually some form of iron oxide. The removal efficiencies of these medias are greatly influenced by pH. Generally, the lower the pH, the greater the capacity throughput. An arsenic selective media will provide a tremendous throughput capacity at pH 6.5, a moderate capacity at pH 7.5 and an uneconomical return at pH 8.5. This is because at lower pH, ions that compete with the iron oxide adsorption space are not as viable. For instance, silica is not very well ionized at pH 6.5 and yet is partly ionized at pH 8.5 and can consume capacity rapidly.
Using strong base anion resins to adjust pH
Can a strong base anion resin be used in a configuration that would allow it to be used as a pH adjustment device? Most systems that are removing trace contaminants benefit from the lead/lad process scheme where two units are placed in series. The first unit is the “worker” and the second unit is the “polisher.” What if we installed a strong base anion unit in the “worker” position and an arsenic selective media in the “polisher” position? The strong base anion unit would be run until some pre-determined point before alkalinity breakthrough, operating as a dealkalizer, thereby delivering a feed to the arsenic selective unit of a lower pH water. The anion would do the bulk of the work and the selective media would remove any traces of arsenic while operating under optimum (relatively low pH) conditions.
This theoretical approach would be limited to systems exhibiting:
- Low to moderate levels of TDS
- Influent water quality that is not variable
- Ability to constantly monitor operation.
Care must be taken to ensure continuous and proper operation of the lead unit. If the lead unit should fail (brine tank not full, unit over-runs or regeneration does not occur when it should), then the effluent from the lead unit would leak alkalinity and the pH would rise. In this upset conditions, the pH rise would for a brief period exceed the influent pH and then would trend down until the pH in equals the pH out.
If the arsenic selective media has been in service as a polisher for some time and is partially exhausted, exposing the media to an influent which has a higher pH than the media has been previously exposed to will cause the media to come into equilibrium with the new pH. This shift in equilibrium can cause a shedding of arsenic that was previously collected, certainly a condition which must be avoided at all costs.
This system therefore requires a close watch on its operations and perhaps warrants the installation of an alarm for brine tank level, monitoring and alarm of pH level on the effluent of the worker unit and flow totalizing alarm on the strong base anion unit.
When considering this type of system, the water analysis provided as the sizing basis must include pH, TDS, sulfate, nitrate, chloride, alkalinity and the contaminant ions of concern. If there is no carbon dioxide level given in the analysis, it must be calculated from the pH and alkalinity measurements. It is also a good idea to consider the hardness level in the water. If there is no softener upstream of the system, it may be necessary to at least have a softened water supply available for the brine dilution and slow rinse of the strong base anion unit.
The theoretical system being proposed here might also prove useful when trying to remove other trace contaminants such as fluoride. Activated alumina medias, which are selective for fluoride, also operate best at lower pH and lower alkalinity.
Frank DeSilva is national sales manager with resin manufacturer ResinTech Inc., West Berlin, NJ.