Ultraviolet (UV) light is produced by an electric arc struck in mercury, or more recently, xenon vapor much like ordinary florescent bulbs. The UV spectrum runs from 100 and 400 nanometers (nm) with the optimal wavelength for bacterial disinfection occurring between 200 and 280 nm.
It has been in use to disinfect water in the U.S. since 1916 and the Department of Health, Education and Welfare (DHEW) issued guidelines in 1966. Since then, the standards have been changing as research reveals how UV works on various microbes.
As with any disinfectant, the process consists of dose and contact time with different organisms being affected differently. The dose for UV is expressed in millijoules per square centimeter (mJ/cm2). To increase the dose, you can increase the output of the bulb, decrease the distance to the bulb, increase the transmittance of the water or slow the flow through the reaction chamber.
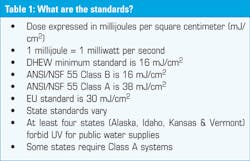
The DHEW set a minimum standard of 16 mJ/cm2 and the NSF standard for Class B units is also 16. The NSF standard for Class A systems is 38, the EU standard is 30 and the state standards vary with at least four states banning the use of UV in public water supplies. See Table 1.
NSF Class A systems are intended for waters assumed to be contaminated and they have other features to restrict the passing of water that has not been properly disinfected. Class B systems are for control of non-pathogenic, nuisance organisms only. Using a non-certified or Class B unit for pathogens is a common misapplication of UV.
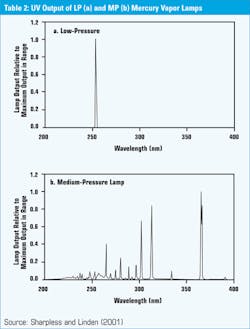
Another misapplication comes from not understanding the outputs of different bulbs. While several types of bulbs are currently being used, the most common are the low (LP) and medium pressure (MP) mercury vapor bulbs. These bulbs have very different outputs as shown in Table 2. The LP bulb has a narrow spike near the 254 nm wavelength, while the MP bulb puts out light across a wider spectrum with a spike between 250 and 300 nm, which is outside what is absorbed by DNA. This is where newer research has shown that proteins which play a role in reactivation are affected.
DNA structure and UV damages
To understand this better, we must take a brief look at the DNA structure and the way UV damages the nucleic molecule. DNA and RNA are the two forms of nucleic acid that regulate cell metabolism and reproduction. They consist of single or double stranded polymers comprised of the basic building blocks called nucleotides. In DNA, nucleotides are broken down further into purines (adenine and guanine) and pyrimidines (thymine and cytosine). In RNA the purines are the same but the pyrimidines are uracil and cytosine. Both purines and pyrimidines absorb UV light, but the rate of UV induced damage is greater in the pyrimidines.
Pyrimidine dimers form when covalent bonds are present between adjacent pyrimidines on the same strand and they are the most common type of damage. Protein-DNA cross-links are covalent bonds between a protein and a DNA strand and they may be important to the disinfection of organisms with double stranded DNA.
Reactivation and dark repair
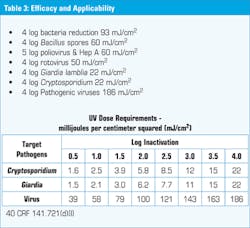
Table 3 shows the dose requirements to inactivate certain target pathogens in water. As you can see, the Environmental Protection Agency (EPA) considers viruses to be particularly difficult to inactivate when taking into account the uncertainty of the hydraulic effects of the UV installation and the possibility of reactivation.
There are three types of reactivation: Photo-reactivation, dark repair and chemical reactivation.
Photo reactivation occurs when a microorganism is exposed to visible light shortly after being irradiated with UV, if certain enzymes are present. The faster the organism reproduces, the faster it can repair the DNA and the more intense the visible light is and the warmer it is while being exposed to the visible light, the better the effect. On the other hand, if the microbes are kept in the dark for at least two hours, few of them will reactivate.
Dark repair refers to all repair processes that do not require visible light, but it does not have to be in the dark. With most bacteria and protozoa, the enzymes begin repair immediately. The bacteria and the viruses that lack the necessary enzymes may have been living inside other organisms that do have it and they can use the enzyme from the host cells. The same is true for parasites in cryptosporidium. The crypto may not be infective, but the parasite becomes the problem.
Chemical reactivation requires the presence of the coenzyme A or nucleic acid nicotineamide-adanine-dinucleotide, or NAD in the water. These molecules are showing up in surface and groundwater and, therefore in tap water, so UV studies have been conducted to determine how they affect the disinfection process. For that reason, public water suppliers using UV are required to use a second agent to ensure that chemical reactivation does not occur.
It is necessary to differentiate between activity and infectivity because there has been a lot of confusion about the ability of crypto and giardia to quickly repair their DNA in the lab. Until 1996, the EPA set a minimum dose for giardia at 180 mJ/cm2 and crypto at 8,700 mJ, because it was assumed that they were infective because they had resumed reproductive activity. In 2001, new studies showed that while the DNA had been repaired, infectivity in humans had not been restored after being exposed to doses as low as 20 mJ/cm2.
Beyond reactivation, there are certain viruses with double stranded DNA that are UV-resistant in the monochromatic 254 mn wavelength produced by LP lamps. Recent research with polychromatic MP lamps show increased inactivation with doses in the 40 mJ range. Additional studies are being conducted to determine if dark repair reactivation translates to infectivity in a human host or if the same laboratory anomaly as with cryptosporidium is at work. In either case, MP UV and pulsed UV have been found to achieve the target 4 log inactivation with doses as low as 40 mJ in the lab.
Increasing effectiveness
There are ways to increase the effectiveness of UV without increasing the power consumption, including adding a catalyst such as titanium dioxide, modulating or pulsing the UV or using very high intensity LED bulbs that have a much more precise wavelength and much lower power consumption, but these methods carry a higher capital cost and are still being researched.
Even with the right bulb there are hydraulic factors that affect the transmittance inside the reactor vessel.
The ideal condition is to have straight through flow with no dead spots or dispersion. There should be no bubbles and every microbe should get a full dose. This can be difficult to achieve in some installations but there are some common mistakes that can be avoided.
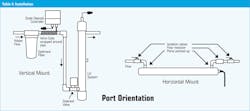
To begin with, the proper orientation of the ports and the location of the flow restrictor can minimize turbulence. It is important to locate the output port at the highest point in a vertical mount installation, or pointing up in a horizontal mount, as shown in Table 4.
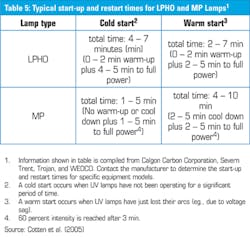
Another mistake is to fit a pressure or flow switch in the output pipe to shut off the lamp when no one is using water. Starting more than four times a day can cause problems with some bulbs and the low and medium pressure mercury lamps likely to be used take time to get to full output (see Table 5) so you could be delivering water that didn’t get a full dose.
Problems caused by UV
Another problem to avoid is the detrimental effect of UV on certain new piping systems. PEX piping is particularly weakened by exposure to UV light so add a leader or an elbow of PVC or stainless before connecting to PEX.
There are also physical factors affecting the transmittance of the water such as suspended solids or turbidity. Pretreatment is necessary to remove suspended solids down to 5 microns and reduce turbidity to something under 1 NTU, preferably 0.5.
Another physical factor that causes problems in UV disinfection is hardness. Scaling on the crystal sleeve inside the reaction chamber and constant rigorous cleaning causes the crystal to lose transparency and reduce the effective dose delivered. De-scaling or softening equipment is often left out of the pretreatment and the savings are more than made up in down time, especially with hardness higher than 200 ppm.
Regular maintenance is just as important as installation when it comes to reliability. Any system will need to be monitored to some extent. Class A systems are prone to false alarms when the water quality varies or voltage is low, so they take a little closer watch. Just being aware of seasonal or transient changes, cleaning the sleeve and replacing the lamp on time will go a long way to keeping a system online and effective. Replace the sleeve about every five years and, if your system is over 10 years old, replace the power supply. UV bulbs are sensitive to voltage fluctuations and power supplies lose accuracy over time.
Advantages and disadvantages of UV
Ultraviolet has a lot of advantages when compared to other disinfection options. The systems are easy to install, there are no chemicals to mix or store, there is no danger of overdose and there are no known toxic byproducts. UV is not affected by pH or temperature and requires only a short contact time making the system simple to operate.
The main disadvantage is that there is no residual, which means contamination resident in the downstream piping is not affected and that water disinfected by UV should not be stored for long periods. Whether new construction or retrofit, all downstream piping should be sanitized and then flushed out with the disinfected water from the system.
Another disadvantage is the difficulty in confirming the efficacy of the UV to inactivate the target microorganisms. The currently-used indicator microbe, E.coli, is very susceptible to inactivation so its absence in a sample will not tell the whole story. It is better to use a heterotrophic plate count (HTC), which includes a variety of microbes and some parasites, but not crypto or giardia. Those two microbes take a separate test that can take two weeks and cost over $300 at a good lab. Continuous monitoring is virtually impossible.
Conclusion
UV will become an even more powerful disinfection tool as researchers learn more about how the various microbes are affected, and manufacturers reduce the overall cost and power consumption of the lamps while zeroing in on the appropriate wavelengths. We need to explore the protein-DNA cross-link and how to fully take advantage of it in UV-resistant viruses. The trend away from mercury vapor bulbs is a welcome improvement in the environmental impact of UV systems. It should also lead to longer-lasting, more powerful and energy-efficient lamps with more precise outputs.
Meanwhile, UV is at work disinfecting drinking water from large scale municipal plants all the way to a single cup on a back woods trail. And each step of the way there are no chemicals or disinfection byproducts added to the water to make it safe. This technology is advancing along with ozone and Nano-alumina as viable options to deal with chlorine-resistant microbes as they arise.
With so much of the country suffering from drought, the bigger question may be where we are going to get the water to treat. So much attention is being paid to the energy side of the equation, there seems to be little being said of our water crisis. Water efficiencies and conservation from alternative sources, such as rainwater and recycled water, will make disinfection technologies even more valuable in the future.