Oil Refining and Petrochemical Wastewater Treatment: Creating a Better Mousetrap
For approximately 70 years now, biological wastewater treatment has been the workhorse for industrial wastewater treatment. Most industrial manufacturing facilities generating substantial volumes of wastewater deploy some form of biological treatment technology if they need reductions in organics, nitrogen, and sulfur.
In the oil refining industry, a wide range of hydrocarbon species and lots of free oil add substantially to the organics loading. In the petrochemical industry, the range of complex hydrocarbons is even greater, although there are generally far lower levels of free oil.
Many engineers believe that these two industrial wastewaters are the most difficult to treat using biological treatment because there are many recalcitrant and inhibitory compounds in the wastewater influent. As such — and especially in these two industries — the overwhelming choice of biological treatment is activated sludge (AS) because of its flexibility for process control in the face of influent concentration variability.
To date, for example, AS is the only biological design configuration in refining and petrochemical wastewater that has successfully utilized biokinetic modeling tools for precise process control at the full-scale operating plant level. It is this degree of difficulty for bioreactor control in these two industries that has motivated many to investigate treatment alternatives for improved performance.
Background on Electrochemical Advanced Oxidation Process
Advanced oxidation process (AOP) wastewater treatment technologies consist of a family of different processes that have one thing in common: the generation of hydroxyl radicals, one of the strongest oxidizers known to man, second only to fluorine. The range of processes in this family deploy a variety of combinations and configurations using ultraviolet (UV) light and hydrogen peroxide, hydrogen peroxide and ozone, catalytic ozone, ultrasound, cavitation, eBeams, plasma arcs, and electricity, to name a few. The first acclaimed AOP was Fenton’s Reagent.
It is important to note that AOPs are markedly different from simple oxidizers such as chlorine, peroxide, permanganate, UV light by itself, etc., which are much weaker oxidizers than the hydroxyl radical. Electrochemical AOPs (eAOP) generate hydroxyl radicals from electrochemical reactions on the surface of the semiconductor anode in an electrochemical cell. In short, when electrons leave the anode, a hole in the valence band of the semiconductor then reacts with water to create hydroxyl radicals.
Over the last five years, several of the AOP technologies have been pilot tested for refinery and petrochemical wastewater applications both at the central wastewater treatment plant (WWTP) as well as for upstream pretreatment. Most these trials included ozone as part of AOP design.
Most of these attempts failed for the same reason: The temperature of these wastewater streams was too high, and as such the cost of ozone was too high. A few successes were observed when the wastewater was cooled.
The AOPs that used hydrogen peroxide also experienced high failure rates in these same applications, mainly because of the wide range of competing reaction species present in these wastewater streams. Interestingly Fenton’s Reagent has produced impressive contaminant reduction results in these applications, but the operational expense (OPEX) was much greater than biological treatment. AOPs that use eBeams and plasma arcs have not yet been tried in the refinery and petrochemical applications because more research and pilot tests need to be conducted.
Around seven years ago, eAOPs were tested on refinery and petrochemical wastewaters using two-dimensional electrodes. The 2-D eAOPs had a reasonable electricity cost but OPEX was prohibitive (much like Fenton’s AOP when compared to biological treatment). Even when the refinery and petrochemical wastewaters had very high chemical oxygen demand (COD) to biological oxygen demand (BOD) ratios, the 2-D electrode still had about 25 percent higher electrical cost than the cost of aeration in AS treatment.
Around three years ago, eAOP technology advanced again with the development of three-dimensional electrodes in which the wastewater flows through the electrodes. This advancement greatly increased the electrode surface area per volume of reactor and the mass transfer rate. Consequently, the electricity usage to drive the hydroxyl radical reaction dropped dramatically. Currently, the electricity cost for eAOP appears to be about 15 percent lower than the electricity cost of aeration in an activated sludge treatment plant.
This stand-alone advantage is just the beginning of the benefits of eAOP, which also include:
- A far greater range of tolerance to temperature compared to biological treatment and other AOPs. It can directly treat wastewater up to approximately 200˚F.
- No residual waste stream, only effluent, so there is no disposal of biomass, sludges, regenerants, precipitants, etc.
- Only one OPEX cost, electricity, which also is usually plentiful and cheap in refineries and petrochemical plants since electricity is typically generated onsite.
- One maintenance cost: periodic cleaning of the electrodes.
- A short hydraulic retention time (HRT) for nearly complete oxidation of the wastewater contaminants: minutes versus hours as compared to biological treatment. Consequently, the eAOP system footprint is smaller by comparison.
- A near linear stoichiometric relationship between electricity usage and contaminant removal, and it can be dialed up or down depending on the wastewater composition and concentration. In contrast, many other treatment technologies have variable hyperbolic effectiveness with respect to destroying organic contaminants, making it difficult to respond to variable wastewater contaminant concentrations. As such, eAOP performance is much more predictable than many other alternatives.
- Extreme ease of operation because it only has three process variables, making process control much simpler than most alternatives such as biological treatment. Because of this, the cost of process control analytical testing and related manpower are greatly reduced as well.
For petrochemical wastewater, eAOP technology looks to have arrived as a very attractive complete replacement for biological treatment. In this application, eAOP can outcompete biological treatment because:
- Petrochem WWTP influent contains many organic contaminants that are either recalcitrant or even inhibitory to biomass growth, but they are very oxidizable using eAOP. The wide range of organic species present in petrochemical wastewater poses no problems such as incomplete or partial oxidation, which is often found with biological treatment.
- Petrochem WWTP influent contains little to no total suspended solids (TSS) or oil and gas (O&G), so there is little potential for electrode fouling in the eAOP reactor.
- Petrochem WWTP influent is relatively stable with respect to variation in contaminant levels when compared to oil refining influent. However, dynamic influent variability can be tolerated with eAOP because of the ease of process control adjustments when compared to biological treatment.
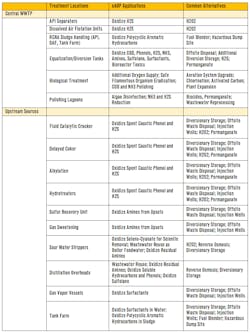
For oil refinery wastewater, eAOP presents a number of attractive options for upstream source pretreatment. Particularly for troublesome streams that could cause problems downstream in the central WWTP, and especially for the protection of the AS biomass, eAOP upstream pretreatment would be of great interest. A summary of potential applications for upstream wastewater source control and pretreatment with eAOP are presented in Table 1.
In the near future, eAOP may likely also outcompete and replace biological treatment in oil refinery wastewater. This will be possible when refineries are able to remove O&G and TSS reliably and consistently before the wastewater enters the eAOP reactor. When TSS and O&G excursions occur, fouling of the electrodes will often cause eAOP reactors to fail, similar to how membrane fouling often causes membrane bioreactors to fail.
A Case History: eAOP vs. AS in Petrochemical Wastewater Plant
Ninety-five percent of the U.S. production of ethylene takes place in Texas and Louisiana. One of the top five largest ethylene and related products manufacturers in Texas is located in Baytown. The amount of ethylene production is generally proportional to the amount of wastewater that is generated.
Unlike oil refining, this type of petrochemical plant generates wastewater that is virtually free of oil and grease and suspended solids. However, it usually has a much wider range of biologically recalcitrant and inhibitory molecules, and thus the degree of stress on the AS biomass is usually much greater than in oil refining wastewater. These were important considerations in the justification to perform a field pilot study comparing the existing AS process to eAOP. The eAOP field pilot was run on a side stream from the AS on a continuous feed basis and was subsequently compared to the full-scale AS.
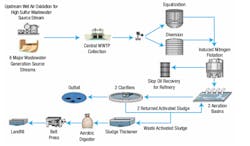
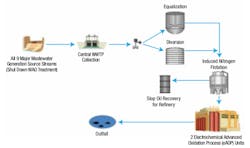
The petrochemical plant where this field pilot study was performed generates around 1,500 GPM of wastewater. Figure 1 illustrates the process flow diagram (PFD) of the existing WWTP. Figure 2 illustrates the PFD of the projected and potentially scaled-up eAOP system.
The eAOP field pilot reactor for this study consisted of a 5-gallon vessel with an operating wastewater capacity of 4.5 gallons. The continuous flow rate through this pilot reactor was approximately 200 mL/min, resulting in an average HRT of about 1.4 hours.
The eAOP reactor was a once-through reactor. The cathode and anode flow-through 3-D electrode assemblies (proprietary) were fully submerged in the vessel, and standard electrical outlets powered them. The continuous feed to the pilot reactor was a side stream that was fed to the full-scale AS influent just after the induced nitrogen flotation (INF) effluent.
It is important to note that during the pilot study, one upstream source of the nine total wastewater source trunk lines contained very high H2S levels and was typically pretreated with wet air oxidation (WAO), another type of AOP. The average WWTP influent contaminant levels of concern to both the AS and the eAOP over this 6-week period were:
- COD: ~1,100 ppm
- Free NH3: ~85 ppm
- TKN: ~240 ppm
- BOD: ~550 ppm
This continuous feed pilot study was conducted over a 6-week period, during which 72 data points were collected to compare the design considerations illustrated in Figures 1 and 2 against the following parameters:
- Full-scale AS contaminant removal vs. pilot eAOP contaminant removal
- Full-scale AS HRT vs. pilot eAOP HRT
- Full-scale AS aeration electricity consumption vs. pilot eAOP electrode electricity consumption
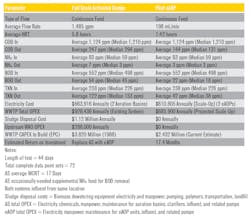
Table 2 summarizes the results of this eAOP pilot study and compares the performance, the OPEX, and the CAPEX considerations of the eAOP design versus the existing AS.
Pilot Study Conclusions
In summary, this field pilot study marked the first time in history that any technology substantially outperformed biological wastewater treatment in petrochemical wastewater on the basis of the removal rates of the contaminants of concern, the OPEX, and the CAPEX.
The projected CAPEX for this scaled-up eAOP design came in around 36 percent lower than that of the original design of the existing AS, mainly based on these considerations:
1. The two required full-scale eAOP reactors for this facility essentially replace the two existing AS aeration basins, both systems in parallel. Since eAOP does not produce any sludge byproducts, the remaining major AS equipment components below are not needed:
- Clarifiers (2)
- Sludge thickener (1)
- Aerobic digester (1)
- Belt press (1)
2. The footprint and thus the land use of the eAOP system is radically smaller than that of the AS, because:
- The HRT required for eAOP Reactors was around 75 percent lower than that of the existing AS.
- The eAOP system eliminates the need for the other AS sludge handling components.
The contaminant removal that was observed for the pilot study showed that the eAOP reactor not only removed all four of the major contaminant groups of concern around 4.5 times faster than the AS but also produced lower final effluent levels for all four contaminants.
The OPEX comparison between the eAOP Field Pilot and the existing AS during this study was dramatic, as summarized below:
- Electricity cost that is based on the eAOP reactor performance was around 8 percent lower than that of the AS aeration basins.
- Electricity cost over the entire WWTP when considering the shutdown of the AS sludge handling equipment and related pumping was around 42 percent lower with eAOP as compared to AS.
- The projected annual savings of no biomass sludge handling treatments and no subsequent offsite landfill disposal was around $1.1 million per year. This figure included the elimination of polymer additions to the clarifiers and at the belt press.
- Substantial OPEX reductions were projected for maintenance costs, mainly because the eAOP system has essentially no moving parts, whereas the AS has many moving parts in numerous locations that result in substantial routine downtime. Periodic electrode cleaning is the only significant eAOP maintenance cost and is relatively straightforward and simple. Consequently, eAOP essentially has little downtime. Downtime is another expense for AS because it leads to lost production.
- Additional smaller OPEX reductions were also projected for pH control chemicals since eAOP has a much wider pH control operation range than that of biological oxidation and especially for nitrification of NH3.
- Additional smaller OPEX reductions were also projected in process control costs for analytical testing and manpower when considering that eAOP has only three process variables versus dozens for the AS.
- This pilot study did not evaluate the impact of shutting down the WAO that pretreats one of the nine upstream wastewater sources, which contains a high concentration of H2S. Currently, WAO is used to treat this steam because H2S pretreatment is required to protect the downstream WWTP biomass. However, H2S is one of the first species to be completely oxidized with AOP. This WAO has a very high OPEX, being an order of magnitude higher in OPEX than the eAOP. In addition, this WAO has a long history of frequent maintenance and downtime. As such, it is anticipated that the eAOP replacement of the AS would have an additional major OPEX reduction because the WAO could be shut down.
Perhaps the most significant feedback regarding the conclusion of this pilot study came directly from the client, who noted that "even if the cost of eAOP was higher than biological treatment, we would still be very interested in the replacement because the process control is so much simpler and reliable." WT
Authors' Note: Refinery Water Engineering & Associates (RWE) is a consulting engineering firm that specializes in oil refinery and petrochemical wastewater optimization for its WWTP clients. RWE is not aligned to any technology and is a non-biased third-party evaluator of wastewater treatment technologies. RWE’s is only aligned with its client’s best interests.About the Authors
David G. Kujawski has 42 years of experience in water and wastewater treatment systems engineering, having performed projects in over 200 treatment facilities including 52 U.S. oil refineries and 13 Latin America oil refineries. He founded Refinery Water Engineering & Associates Inc. (RWE), a small environmental engineering firm uniquely specialized on oil refinery applications, and served as its COO for twelve years. Kujawski earned a B.S. in chemistry at St. John’s University and a process engineering degree from Chevron’s Engineering Certification Program, which included graduate work at the University of Texas and UCLA.
John C. Crittenden, Ph.D., P.E., F. ASCE, NAE, CAE, is the director of the Brook Byers Institute for Sustainable Systems, Hightower Chair and Georgia Research Alliance Eminent Scholar in Sustainable Systems at the Georgia Institute of Technology. He is a member of the U.S. and Chinese National Academies of Engineering and was selected as one of the 100 Pre-Eminent Chemical Engineers of Modern Times by AIChE. The American Society of Civil Engineers named Dr. Crittenden the recipient of the 2020 Simon W. Freese Environmental Engineering Award and Lecture, which recognizes “extraordinary accomplishments in using fundamental scientific principles and current research findings to solve the most challenging water quality problems.”
About the Author

David Kujawski
David G. Kujawski has 42 years of experience in water and wastewater treatment systems engineering, having performed projects in over 200 treatment facilities including 52 U.S. oil refineries and 13 Latin America oil refineries. He founded Refinery Water Engineering & Associates Inc. (RWE), a small environmental engineering firm uniquely specialized on oil refinery applications, and served as its COO for twelve years. Kujawski earned a B.S. in chemistry at St. John’s University and a process engineering degree from Chevron’s Engineering Certification Program, which included graduate work at the University of Texas and UCLA.

John Crittenden
John C. Crittenden, Ph.D., P.E., F. ASCE, NAE, CAE, is the director of the Brook Byers Institute for Sustainable Systems, Hightower Chair and Georgia Research Alliance Eminent Scholar in Sustainable Systems at the Georgia Institute of Technology. He is a member of the U.S. and Chinese National Academies of Engineering and was selected as one of the 100 Pre-Eminent Chemical Engineers of Modern Times by AIChE. The American Society of Civil Engineers named Dr. Crittenden the recipient of the 2020 Simon W. Freese Environmental Engineering Award and Lecture, which recognizes “extraordinary accomplishments in using fundamental scientific principles and current research findings to solve the most challenging water quality problems.”