Oxidative Evaporation Process and Its Application in Treating Spent Casutic
by Philip T. Chen, PhD, PE
• Ideal for refinery, petrochemical, electroplating, pharmaceutical and ultra-pure water facilities, a new industrial waste treatment method destroys COD in wastewater and brings non-reactive in solution to saturation for residue recovery in solid form.
The basic concept of wastewater treatment can be simply put as the conversion of contaminants characterized as chemical oxygen demand (COD) into substances of non-COD via application of an oxidizing agent, such as oxygen gas. Ideally, carbon dioxide (CO2) and water (H2O) would be the desirable final products in waste treatment; yet, many incompletely oxidized compounds or compounds resistant to oxidative reaction remain in the water solution. Those compounds, categorized as low oxygen demand substances, are the most difficult to remove from water being treated.
Oxidative methods used in the field of wastewater treating include:
- Application of strong oxidizing agents, such as permanganate, chromate, ozone, hydrogen peroxide activated by UV or hydro-oxy radical, etc., to chemically oxidize contaminants in water.
- Organic compounds are decomposed or assimilated into bacterial cells at the expense of oxygen in maintaining the biological cell activities.
- Organic contaminants are destroyed by rapid oxidation through high temperature gasification/combustion, i.e., incineration.
In addition, in the 1960s, a liquid phase oxidation process, commonly called Wet Air Oxidation (WAO), was developed and has been widely used since in treating industrial effluent. The Oxidative Evaporation (O-E) Process, discussed in this article, is a modified WAO process in which organic contaminants are destroyed in liquid phase with use of oxygen under high temperature and pressure, while residues (i.e., the low oxygen demand substances) are recovered from water in solid form through evaporation of water at the same time.
Many hazardous wastes containing pesticides, cyanides, phenolics and sulfur compounds are the most difficult to treat with conventional methods. Many industries face difficulties in finding adequate means to dispose of these wastes economically. The O-E Process is suited for treating such difficult wastewater.
Conventional Treatment
Spent caustic solution is a most obnoxious aqueous waste stream generated in manufacturing plants and refineries producing ethylene and petrochemicals. It’s a caustic solution used in scrubbing thermally cracked gas products. The spent caustic is loaded with hydrogen sulfide (H2S), disulfides, phenolics, mercaptans and short-chain hydrocarbons that are generally termed as polymer precursors. It appears generally as a dark brownish solution with a pungent characteristic smell of olefins.
Commercially available technologies currently used for treating spent caustic solution include the following:
- Neutralization-Steam stripping (Lo-Cat process) – mainly for recovery of H2S, leaving organic and salt untreated. Neutralized effluent requires further treatment at the end-of-pipe.
- Neutralization-Air stripping (Shell International Chemie Masstschappij or SICM process and Nippon Petrochemical process) – aimed at converting H2S to thiosulfate to reduce its odor. Treated effluent also needs treatment at the end-of-pipe for the same reason as the first technology.
- WAO (represented by Siemens Zimpro process) – utilizes compressed air at high temperature and pressure to oxidize H2S to sulfate, and organic components to carbonates, yielding a salt laden solution as an effluent that also contains organic residues. Further treatment is required for removing the residual organic compounds and salts.
Oxidative Evaporation
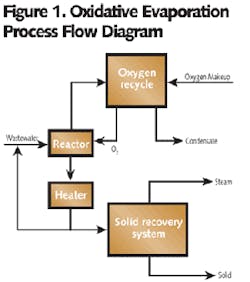
The O-E Process is similar to WAO technology in that the oxidation reactions of organic constituents and sulfides are carried out at an elevated temperature and pressure. However, heat generated in the oxidative process reactions is utilized to evaporate water in the reactor (see Figure 1).
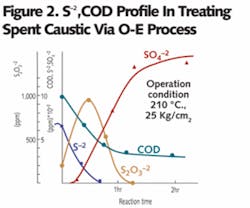
Spent caustic is an alkaline solution with a high COD, sulfides and salt content. These constituents react with oxygen under high temperature and pressure to yield carbonates and sulfates, which are retained in the water phase in the reactor. The concentration profile of these constituents in the reactor is shown in Figure 2. The profile shows that conversion of sulfides and organic compounds into sulfates and carbonates is completed in two hours under the given condition. As a result of treatment, water is evaporated as low pressure steam and the organic constituents are converted into carbonates/sulfates, which are removed from solution in solid forms.
The COD values of spent caustic vary widely according to the sources. Generally, it’s in the range of 10,000-40,000 ppm. With the O-E Process, the efficiency of the COD removal is invariably higher than 90% for operations with temperatures higher than 200ºC. The process performance in removing COD appears to be quite sensitive to operating temperature (see Table1).
The O-E vs. WAO Process
1. The process performance in treating spent caustic – For conventional WAO processes, the process flow is maintained as two-phase flow. The oxygen gas or air is injected into the liquid and they flow through the reactor co-currently. To maintain two-phase flow, evaporation of water must be suppressed and the system must, therefore, be maintained at high pressure throughout the process. Consequently, final effluent is a mixture of liquid and gas.
In the O-E Process, the flow is maintained as a single-phase flow after the reactor. In the reactor, O2 gas and liquid are brought into contact counter-currently; the process reactions and evaporation of water take place simultaneously. Vapor generated is separated from liquid in the reactor. The liquid from the reactor bottoms is further evaporated to recover salts in solid form.
Both processes are equally effective in reducing COD in the spent caustic solution; however, even with high COD removal, the residual COD – in the order of 1000s of ppm, which represents the COD in the effluent of the reactor – remains too high for final disposal. In the WAO process, this residual COD along with the salts in the effluent needs to be further treated at a secondary treatment facility. In the O-E Process, the residual COD is recycled back to the reactor for re-processing. Salts retained in solution, mostly in the form of carbonates/sulfates, are concentrated through evaporation of the water and recovered in solid form for final disposal.
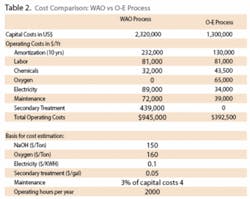
2. The cost comparisons – Annual plant operating costs for both processes are estimated as shown in Table 2. By converting the treatment cost into per ton basis of treatment, the O-E plant is $26/ton, with $62/ton for WAO.
Varied Industrial Applications
The O-E Process is suited for many applications other than treating spent caustic in manufacturing industries. In general, it can be used to treat effluent with a high organic COD content and high concentration of salts, especially when the organic content is toxic in nature or the salts contain heavy metals.
1. Applicable industries – The O-E Process can be designed to operate under different conditions according to requirements of the specific wastewater to be treated. The design also includes allowance for processing colloidal or suspended matter in the feed water. Every process effluent is unique in nature. They’re treated under temperature/pressure conditions specific to the waste to be treated.
2. Prefabricated package plant options – Generally, the effluent stream from manufacturing processes carries a high concentration of organic contaminants and salt content, but flow is relatively small. The O-E Process is well adapted to handle this type of waste stream and can be readily constructed as a packaged unit. The process is practically self-contained: it invariably yields steam condensate and carbonate salts in solid form as the final products. The steam condensate can be reused as process water and the solids can be disposed off-line.
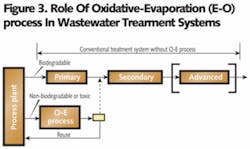
3. Use in conjunction with conventional wastewater treatment systems – The O-E Process can be a standalone waste treatment system or integrated into conventional treatment systems as a pretreatment measure. When it’s used in conjunction with a conventional waste treatment system, the manufacturing plant effluents can be segregated into two streams: 1) a biodegradable stream, and 2) a non-biodegradable or hard-to-treat stream. The O-E plant treats the non-biodegradable/hard-to-treat stream and the biodegradable stream is treated by conventional treatment systems (see Figure 3).
The O-E process eliminates the hard-to-treat compounds and, at the same time, removes salts from the waste solution at the source. By accomplishing these two important goals, it improves the overall efficiency of the conventional treatment systems.
Conclusion
The O-E Process is specially designed for treating difficult effluent streams. It oxidizes organic contaminants to a maximal extent; and residues, including non-oxidized and inorganic salts, are concentrated by evaporating water. In the process, the reaction heat of oxidation is fully utilized for evaporating water, thus minimizing requirements for supplemental energy. As such, this process can provide a total treatment of wastewater by recovering water as steam for reuse and recovering dissolved residues in solid forms, minimizing the volume of waste for final disposal.
About the Author: Based in Montvale, NJ, Philip T. Chen, principal of Resources Engineering Consultants Ltd., is a chemical engineer specializing in process design engineering. With over 30 years of professional experience, he holds numerous U.S. and foreign patents and is the original author of the Oxidative Evaporation Process technology. Contact: 201-573-9346 or [email protected]