Professor POU/POE: PFOA & PFOS in drinking water
Q: What are PFOA and PFOS, what are their health concerns, and how do they get into drinking water?
A: Both are polyfluorinated organic chemicals that have many commercial uses and are widely distributed in foods and the environment at low concentrations. They are difficult to treat and are not biodegradable, so they have been detected at low levels in wastewater and some drinking water. Very high detection frequencies are found in human blood.
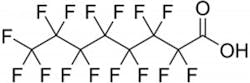
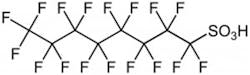
PFOA is perfluorooctanoic acid (C7F15COOH), and PFOS is perfluorooctanesulfonic acid (C8F17SO3H). Numerous perfluorinated compounds are in the environment, including commercial products and byproducts and wastes released from production facilities. PFOA and PFOS are the most widely produced perfluorocarbons worldwide.
PFOA
They both contain eight carbon atoms, mostly in a linear chain. The per- prefix in the names means that all of the available hydrogen atoms that were attached to carbon atoms have been replaced by fluorine atoms. That causes them to be biologically stable chemicals and to be very lipophobic (repel fats) and hydrophobic (repel water), and it also increases their acidities. They also have low volatility. As surface-active agents, they are used in many industrial and
commercial products, such as water and soil repellant coatings on carpets, fabrics and leather, foams used in firefighting, electroplating, photography, paper coatings, and pesticides. Some PFOA and PFOS compounds can form by degradation from higher molecular weight perfluorocarbons.
PFOS
Physical and chemical properties
Water solubility and biological transport are significantly affected by the cation that is associated with PFOA. The water solubility of the ammonium (NH4+) salt of PFOA is at least 50 times greater than the solubility of the acid form, which is already highly ionized. The water solubility of PFOS salts is about the same as the acid form.
PFOS and related chemicals were in Scotchguard, which is manufactured by 3M, but its production in the U.S. was phased out beginning in 2000. Scotchguard was reformulated in 2003, and PFOS was replaced by perfluorobutanesulfonic acid (PBOS). The half-life of PBOS in humans is about one month versus 5.4 years for PFOS. Existing and imported stocks of PFOS can still be used under more controlled conditions.
PFOA was covered in a U.S. Environmental Protection Agency (EPA) stewardship program that specified that by 2010, eight companies agreed to reduce global emissions from facilities, reduce precursor chemicals that breakdown to PFOA, reduce product content by 95 percent, and work toward eliminating them from emissions and products by 2015. The European Union banned the use of PFOS in products in 2006 (maximum content 0.005 percent by weight), but applications for photolithography, mist suppressants for chromium plating and aviation hydraulic fluids were exempted.Occurrence in humans and animals
Studies between 1999 and 2004 detected PFOA, PFOS and other perfluoro chemicals in 95 to 100 percent of human blood samples. More recent monitoring indicates that the blood levels are declining. Mean serum concentrations in the U.S. general population were 2.08 micrograms per liter (µg/l) for PFOA and 6.31 µg/l for PFOS in 2011 and 2012, down from 5.21 µg/l and 30.4 µg/l, respectively, in 1999 and 2000. People living near a user industrial facility had mean PFOA serum levels of 83.6 µg/l. Workers had mean serum levels of 1,760 µg/l PFOA and 1,320 µg/l of PFOS. Significantly higher levels have been reported in some western countries, and high levels have been detected in some wild animals around the world.
Virtually 100 percent of the ingested PFOS is bound to proteins in rat, monkey and human plasma. Rat studies indicated at least 95 percent of PFOS was absorbed after ingestion, and only 5 percent was eliminated within 48 hours. The elimination half-life of PFOS in humans is on the order of 5.4 years. For PFOA it is 2.3 years, which indicates that they will both accumulate and that the body burden levels cannot decline substantially until intake is significantly reduced. In humans, PFOS has been found to distribute mostly to the liver and blood; It has also been identified in umbilical cord blood and breast milk. In humans, the amount of PFOS in the serum and liver tissue is similar, while in animals the amounts found in the liver are higher than in the serum. Studies in rats indicated that PFOS absorption also occurs by inhalation and dermal exposure.
Human and animal health effects
Many human studies have examined relationships between human blood levels of various perfluoroalkyl compounds and adverse health effects. The Agency for Toxic Substances and Disease Registry (ATSDR) said that interpretations are difficult because the results are not always consistent. The most consistent results indicated relationships may exist between elevated blood cholesterol and high PFOA or PFOS serum levels. Increased uric acid associations may indicate increased risk of high blood pressure. Liver damage possibilities may be indicated by effects observed in animals. However, ATSDR goes on to say that humans and animals often react differently to those chemicals, and not all effects observed in dosed rats and mice may occur in humans. According to ATSDR, no conclusive evidence supports that perfluoroalkyls cause cancer in humans. Although some studies indicated increased cancer risks at high exposure levels, they were inconsistent, and most did not control for confounding factors such as smoking.
A multiyear study near a Dupont facility in West Virginia that manufactured PFOA has produced equivocal correlations. A comprehensive review by Steenland et. al. in 2010 concluded: "Epidemiologic evidence remains limited, and to date data are insufficient to draw a firm conclusion regarding the role of PFOA for any of the diseases of concern." Likewise, a review of pregnancy outcomes by Savitz et. al. in 2009 stated: "The data provide little support for PFOA or PFOS being related to pregnancy outcome, with some uncertainty regarding pre-eclampsia and aggregated birth defects."
Occurrence and exposure in water
Detections of PFOS and PFOA have been reported in numerous locations often in the vicinity of manufacturing and user facilities such as airports where discharges have occurred and soil and water contamination are present. Several other perfluorinated compounds are in common use and have been detected in the environment and in humans, but currently PFOA and PFOS have received the most attention.
The EPA’s Third Unregulated Contaminant Monitoring Rule (UCMR 3) reported data for PFOS and PFOA as well as four other perfluorinated compounds from 35,060 samples in 4,788 public water supplies. The occurrence levels and exposures from drinking water were very low using a very sensitive analytical method, USEPA Method 537. The Minimum Reporting Levels (MRL) for the UCMR were 0.02 µg/l or 20 parts per trillion for PFOS, and 0.04 µg/l or 40 parts per trillion for PFOA. The drinking water Reference Concentrations and provisional short-term health advisories are 0.2 µg/l for PFOS and 0.4 µg/l for PFOA. The other four perfluorinated chemicals were generally in a similar range of occurrence and concentrations, but the EPA did not have a reference concentration for those chemicals.
somechaisom/iStock
Public concerns were recently raised in several locations where groundwater contamination was detected in New Hampshire and Vermont. Levels as high as 0.62 µg/l were reported in some private wells in the vicinity of a plastics manufacturing facility. Several states have issued health advisories usually lower than the EPA’s.
Health-related benchmarks
The EPA released comprehensive PFOA and PFOS Health Effects Documents for peer review in 2014. It proposed 0.03 µg/kg/day (2.1 µg/day) as the reference dose (RfD) for PFOS, and 0.02 µg/kg/day (1.4 µg/day) as the RfD for PFOA. On May 19, 2016, the EPA published lifetime Health Advisories from RfDs of 0.02 µg/kg/day for the most sensitive population, lactating women, and uncertainty (safety) factors of 300 and a drinking water relative source contribution of 20 percent that usually provides some additional margin. It also indicated suggestive evidence for human carcinogenicity. The EPA concluded that drinking water with individual or combined concentrations of PFOA and PFOS below 70 parts per trillion would not cause adverse health effects over a lifetime of exposure. Those levels reflect a significant margin of protection for the most sensitive populations. Also in the findings, the primary source of exposure was from food and consumer products, and was typically a localized issue where a manufacturing plant or airfield made or used these chemicals.
Water treatment technology
Several treatment technologies are capable of removing PFOS and PFOA from drinking water. These include granular activated carbon (GAC), which can be can be thermally reactivated; anion exchange and reverse osmosis (RO); and possibly other membrane filtration. The efficacy of anion exchange processes can be affected by other components in the water such as dissolved solids and organics, and membrane processes can be improved by the addition of other minerals. The waste product concentrates from anion exchange and membranes require appropriate management and should not be released to the environment.
Conclusions
Several early references identify drinking water as a significant source of exposure to PFOS and PFOA, but based on the large-scale UCMR 3 results, that is not valid for public water supplies. Food is the largest contributor to general population exposure, but some exposures may occur from inhalation or dermal contact with consumer products. Declining trends in human exposures are because of bans, phaseouts and product residue restrictions, and these are reflected in lower population blood levels. Whether national drinking water standards are needed or if health advisories are sufficient to protect health is debatable. High exposures to workers in production and manufacturing facilities as well as measurements in some nearby populations are related to the occupational circumstances and local environmental releases.
According to ATSDR, human epidemiological studies of high exposure populations have been generally inconsistent, although cholesterol increases and modest correlations with liver enzymes have been suggested. The EPA concluded suggestive evidence of human cancer risk exists but negligible risk is below the health advisory values.
The new drinking water health advisories for lifetime exposures are 0.07 µg/l (70 parts per trillion) for individual or total PFOA and PFOS, replacing the provisional short-term advisories. These types of chemicals are essentially not metabolized after ingestion or biodegraded in the environment. The long retention times and bioaccumulation after ingestion indicate that the effective exposures are greater than indicated by daily intake levels.Numerous other persistent perfluorinated organic compounds in the environment and human blood warrant controls to minimize human occupational and environmental exposure potential.
Dr. Joe Cotruvo is president of Joseph Cotruvo and Associates, LLC, Water, Environment and Public Health Consultants and technical editor of Water Technology. He is a former director of the EPA Drinking Water Standards Division.