Ensuring Purity at the Source VIa Lab Analysis
by Drs. Shona McSheehy and Martin Nash
• Determination of trivalent and hexavalent chromium in bottled mineral and spring water using HPLC coupled to ICP-MS.
Extensive use of chromium in various industrial processes, as well as erosion of chromium from natural sources, have resulted in widespread occurrence of this chemical in the enVIronment. Monitoring chromium in enVIronmental compartments as well as food and water sources is key to assessing exposure's potential risk. The U.S. EnVIronmental Protection Agency and European Union have specified maximum admissible concentrations of 0.1 and 0.05 mg/L in water for total chromium under their respective drinking water regulations.
Due to significant differences in toxicity, reactiVIty and bioavailability, though, it's also VItal to distinguish between the trivalent (CrIII) and hexavalent (CrVI) states of chromium. CrIII is essential to human life as it plays a VItal role for glucose, protein and fat metabolism with a daily intake of 50-200 µg/d recommended for adults. Acute animal tests have shown CrIII to have moderate toxicity from oral exposure. CrVI, on the other hand, is highly toxic if orally ingested or inhaled due to its strong oxidizing properties. There's eVIdence to suggest CrVI is capable of causing skin, nose, eye and throat irritations in cases of limited exposure, and damage to liver, kidney circulatory, nerve tissues and cancer of the respiratory tract in more extreme cases. For these reasons, the World Health Organization recommends maximum allowable concentrations of 0.05 mg/L for CrVI in drinking water. The use of CrVI is also restricted by the EU Restriction of Hazardous Substances Directive 2002/95/EC.
To assess human health risks from enVIronmental exposure to chromium and chromium species, an accurate analytical method comprising highly sensitive and specific detection is needed. The combination of High Performance Liquid Chromatography (HPLC) and Inductively Coupled Plasma Mass Spectrometry (ICP-MS) proVIdes a VIable solution for rapid, accurate and sensitive determination of CrIII and CrVI species in mineral and spring water.
Experimental
A fully inert Thermo Scientific SpectraSYSTEM™ HPLC pump equipped with an AS3500 autosampler was coupled to the XSERIES 2 ICP-MS using HPLC-ICP-MS coupling pack and HPLC wiring harness – all from Thermo Fisher Scientific. The ICP-MS was operated under standard hot plasma conditions using a one-piece quartz torch with a 1.5 mm ID injector. The spray chamber was cooled to 2°C with the optional cooling deVIce. Integral HPLC and ICP-MS software packages were used in conjunction with an external trigger card to enable automated HPLC accessory control using bi-directional communications and intelligent peak integration facilities. The associated HPLC parameters and analytical conditions for the HPLC-ICP-MS applications are shown in Table 1.
The reversed phase HPLC methodology employed complexation of CrIII with EDTA to improve separation. Due to the carbon containing mobile phase, optional collision cell technology (CCTED) was optimized for the prevention of the polyatomic interference on chromium. The CCT additionally suppresses interference from matrix in the water samples. The analytical methodology was validated using a CRM (BCR CRM-544, lyophilized water) and method limits of detection (LOD) were determined from three times the standard deVIation of species concentrations found in the blank (n=5).
Sample Preparation
Daily working standards were prepared by diluting the appropriate quantity of the commercially available stock solutions (1000 µg.mL-1) of each chromium standard in the HPLC mobile phase. The stock solutions were kept at 4°C in the dark. The lyophilized water was extracted according to the method outlined in the certification report supplied with the CRM. The sample was reconstituted with 20 mL HCO-3/H2CO3 buffer at pH 6.4.
Aliquots of the reconstituted CRM were diluted 1:1 in 20mM EDTA, 2.5 mM TBAP. Mineral and spring water samples were diluted 9:1 in 20 mM EDTA, 2.5 mM TBAP. Spikes of CrIII and CrVI were added to the reconstituted CRM and the mineral water samples prior to dilution with the EDTA solution. Both the standards and samples were placed in a heated water bath at 70°C for one hour to accelerate complexation of the CrIII with EDTA.
Results & Discussion
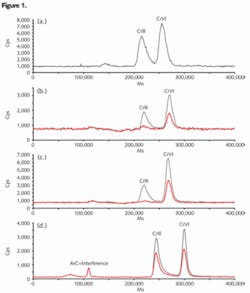
Following the analysis, the chromatographic data was displayed automatically in the PlasmaLab ICP-MS software. An example of the chromatographic separation of chromium-containing standards at a concentration of 1 µg. L-1 is shown in Figure 1 (a). The HPLC methodology using EDTA as complexation agent allowed the baseline separation of CrIII and CrVI with retention times of 215 and 260 seconds respectively.
External calibration curves were generated in the PlasmaLab ICP-MS software using a blank and CrIII and CrVI calibration standards of 0.1, 0.2, 0.5, 1, 2, 5 and 10 µg.L-1. Quantification of CrIII and CrVI species was achieved in several samples using the external calibration curves presented in Figure 2 and fully quantitative data processing was achieved using the software's automated peak integration tools.
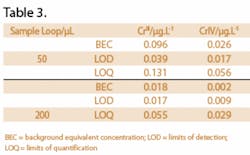
The mineral water samples were selected from a local supermarket and were analyzed in triplicate. Their mineral content and the quantitative data for CrIII and CrVI are presented in Table 2. The chromatograms for samples D and E are presented in Figure 1 (b) and (c) respectively. All the samples contained CrVI as the major species with concentrations varying between 0.054 and 0.409 µg.L-1. Three of the five water samples contained CrIII at levels above the limit of detection but only just above or below the quantification limit. Due to the low levels of chromium species in these samples, the detection and quantification limits of the methodology were improved with a 200 µL sample loop, rather than a 50 or 100 µL sample loop. Figures of merit are presented in Table 3.
The method was validated for the mineral and spring water samples by determining the recovery of CrIII and CrVI standards added to the samples prior to the complexation step (relevant data are presented in Table 2). The recovery determined for the five samples analyzed fell between 90% and 105%, even for spikes at levels as low as 0.1 µg.L-1 of each chromium species. Further method validation was performed through duplicate analyses in two independent bottles of the lyophilized solution. A chromatogram of reconstituted lyophilized water is presented in Figure 1 (d). Due to the higher levels of chromium species in this CRM, a sample loop of 50 µL was used for the fully quantitative calibration and sample.
The chromatogram shows that CrIII and CrVI were found in the CRM. The associated quantitative data is presented in Table 2 and there is satisfactory agreement between the measured and certified values for CrIII and CrVI. The pH of the reconstituted solution, however, is critical for preventing hydrolysis of the CrIII species and the solution should be complexed or analyzed as soon as possible after the recommended purge with CO2.
MDLs and LOQs (limits of quantitation) for CrIII and CrVI species were determined in accordance with the 3s and 10s models respectively using fully quantitative analyses of method blanks (n=5) and the associated figures of merit for sample injection volumes of 50 and 200 µL are presented in Table 3.
Conclusion
CrIII is moderately toxic if orally injested in quantities higher than 200 µg/d. CrVI, on the other hand, is highly toxic if orally ingested or inhaled causing skin, nose, eye and throat irritations as well as damage to liver, kidney circulatory and nerve tissues and cancer of the respiratory. As a consequence, the EPA and EU have specified maximum admissible concentrations for total chromium and chromium species under their respective drinking water regulations. Concentrations must be monitored closely to assess human health risks and ensure regulatory compliance. The combination of HPLC and ICP-MS proVIdes a validated solution for fast, precise and sensitive speciation of CrIII and CrVI species in mineral and spring water.
About the Authors: Dr. Shona McSheehy is an ICP-MS application specialist with Thermo Fisher Scientific Inc. in Bremen, Germany. Dr. Martin Nash is ICP product manager with Thermo Fisher Scientific in Cambridge, UK. Thermo Scientific is part of Thermo Fisher Scientific Inc., which offers a range of analytical instruments as well as laboratory equipment, software, ser VIces, consumables and reagents to enable integrated laboratory workflow solutions. Contact: +1 800-532-4752, [email protected] or www.thermo.com/speciation