Naphthenic Acid Removal in Offshore Oil Production
Global offshore oil production is expected to create approximately 15 billion barrels per year (bbl/ yr) of produced water by 2030, which will require treatment for reuse or disposal (GWI, 2014). The technologies used to treat offshore produced water continue to evolve as operators push for efficiency and regulation drives sustainability. The produced water from heavy oil reserves in Brazil are known for having high acidity, especially in regard to naphthenic acids (NAs). Current changes in Brazilian regulations aim to reduce the impact of produced water, specifically water-soluble organics such as NA, discharged into the sea. However, NAs are problematic for oil producers to remove from water because they are amphiphilic molecules, being distributed between the oil and water phases. One of the challenges facing producers is low awareness and/or understanding of NA, including the processes and technologies used to remove NA from produced water streams as well as produced water analysis methods for detecting NA.
This article provides a summary of NA, including the focus from Brazilian regulatory agencies and the challenges faced in removing NA from produced water. The goal of this article is to supply operators, service companies, and technology providers with an appreciation of NA issues and the knowledge to facilitate cooperation that creates an efficient solution.
A New Class of Molecules
Throughout the history of the oil and gas industry, regulatory mandates have been one force for creating change and driving research. When companies are compelled to follow new rules and regulations, the industry refocuses and increases its understanding of the impacts from such regulatory changes. Usually, this research is driven by the search for the most economical, safe solution. A recent regulatory change in Brazil, which would seem minor to most, has created massive challenges for offshore oil and gas operations in the region.
In 2018, CONAMA (Brazil’s National Environment Council) reviewed the methodology used to calculate the residual oil in produced water discharged into the ocean. Due to the findings in its study, the water analysis method for discharged water was changed, and a new class of hydrocarbon molecules is now recorded as oil within the overboard discharge stream. Petrobras and other producers are now forced to understand this new class of molecules and how to eliminate it from the discharged produced water. In fact, the research into solving the produced water treatment issues is still underway as many operators, service companies, and technology companies search for the most economical method to treat millions of barrels of produced water discharged into the ocean every day.
Quantifying Produced Water & Analysis Methodology
In analytical chemistry, methods used to quantify a chemical are critical in accurately determining its concentration. This is also true for Brazil oil producers and their method to calculate total oil and grease (TOG). In fact, the term TOG can be debated and is best described by stating what analytical method was used to determine it. This limits the ability of most people to understand exactly what the impact is from TOG discharged into the ocean because it requires familiarity with specific analytical chemistry methods.
In Brazil, the amount of TOG is determined as the total hexane extractable material (HEM) that is present in the water sample. In other words, TOG records the compounds that are soluble in hexane. The current TOG method in Brazil is SM 5520B, and is similar to the U.S. EPA method 1664 and ISO 11349. This method of TOG quantification excludes anything that is more volatile than hexane and includes nonhydrocarbon compounds that are extracted by hexane.
The change in Brazil’s analytical methodology was a shift within the SM 5520 procedure from the SM 5520F method to the SM 5520B method. The difference in these methods is one sample preparation step that includes silica gel. Silica gel is used as a clean-up step in SM 4420B to aid in the analysis of TOG; however, the silica gel removed the soluble organic fraction of the HEM that, without the silica gel step, will now be measured and recorded as part of the TOG. Therefore, the new method used to analyze the produced water increases the number of chemical compounds reported in water discharged into the sea. It should be noted that globally TOG analysis methods vary by country and region and each method has its own merits.
The Reason Analysis Methods Matter
The structure of NA is defined as molecules with carboxylic acid functional groups attached to aliphatic or ring structure hydrocarbons.1 The origin of the NA in the oil is the result of thermal degradation and biodegradation. The action of biodegradation is responsible for the increase of acidic compounds. Crude oils with high amounts of NA are found in several fields around the world in Angola, Congo, Nigeria, North Sea, Brazil, Colombia, and Venezuela, and consequently, the associated produced water also has high amounts of NA.
A deeper understanding of the produced water from these fields, and more importantly the residual oils in the produced water, is required to explain the importance of the TOG measurement method change. The residual oil in produced water can be separated into three different categories: dispersed, emulsified, and dissolved. Dispersed oil is the easiest to remove because the oil and water naturally separate. Emulsified oil is harder to remove from produced water but can be managed mostly with the addition of chemicals. Dissolved oil is the hardest to remove because the chemical nature of the dissolved oils and water are similar to each other. Typically, dissolved oil removal can be completed using adsorption media, absorption media, or vaporization/distillation for volatile compounds.
In Brazil, the dissolved oils are a unique class of compounds referred to as NAs. NAs have a boiling point that does not allow for vaporization/distillation as a removal method and NAs have a weak attraction to adsorption medias.
It is important to study the equilibriums involved in NA/oil/water systems. NA include amphiphilic molecules (polar and nonpolar parts) and is often found distributed in both the water and oil phases. The distribution of NA between these two phases correlates mainly with the pH of the system.
To further understand why residual oils like naphthenic acids cannot be removed using traditional methods, we need to refresh our memory of organic chemistry. First, let’s review the TOG method that uses hexane to extract oil compounds. In chemistry, molecules that display similar characteristics prefer to be together. During a TOG analysis, nonpolar hexane extracts nonpolar oil compounds. The same principles are applied to oil and gas process equipment to ensure oil is collected and recovered. Additionally, adsorption media used to remove dissolved oils have oleophilic characteristics, so they act like a solid oil particle that can interact and remove the oil but remain contained inside of a vessel. These principles and techniques have successfully separated oil and water for decades, and optimization of these techniques allows for most produced waters to be discharged with TOG <20 mg/L. However, even with the highest degree of optimization, NA cannot be removed with these traditional techniques.
This begs the question, why are NA measured as oil but cannot be removed like oil?
To understand the NA paradox, the TOG analytical method must be understood. The first steps in the SM 5520B analysis method for TOG detection is reducing the pH to <2. Carboxylic acids are weak acids and undergo changes depending on the pH. More specifically, at a critical point, termed the pKa, weak acids shift from being protonated to deprotonated (i.e., from having a hydrogen atom to not having a hydrogen atom).
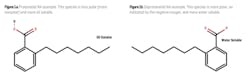
Therefore, in lowering the pH of the water sample the NA molecule goes through a fundamental change. The lower pH causes NA to change from a deprotonated species to a protonated species.2 In the deprotonated form, NAs have a negative charge that increases their water solubility. The protonated form of NA has a hydrogen atom bonded where the negative charge existed, neutralizing the charge or making the NA less polar, and hence less water miscible. Figure 1 shows an example of one naphthenic compound in the protonated form (A) and deprotonated form (B). The added hydrogen atom balances the negative charge and reduces the water solubility while increasing the oil solubility.
The issue of NAs for Brazil is a unique combination of factors that ultimately result in measurement of high concentrations of NA in produced water. Additionally, the NA cannot be removed as a result of the natural pH of the produced water, but they are measured in the SM 5520B analysis method for TOG as a result of both the use of the pH adjustment step and the removal of the silica gel step from the method.
Searching for the NA Solution
Why doesn’t Brazil lower the pH of the produced water to remove the NAs if testing results indicate that lowering the produced water pH would significantly reduce the NA concentration? Two issues are faced when lowering the pH of produced water, one economic and one environmental. The economic issue is simply the cost to lower the pH of thousands of barrels of produced water every day to a more acidic pH of <5, including the logistics of transporting the acid offshore, which is prohibitive and operationally intensive. The environmental issue — and perhaps the more important barrier to lowering the pH — is the potential harm to marine flora and fauna. The toxicity of the water would most likely increase with the lower pH versus the presence of NAs alone.
Although Brazil is only recently starting to explore NA, it has been studied around the globe for some time. Finding the best solution to remove NA from water will require using the collective knowledge the oil and gas industry already has, expertise and collaboration of several water treatment technologies, and the innovative drive of technology, service, and production companies. The optimal solution, which is yet to be discovered, must meet the economic, safety, durability, and size requirements that challenge offshore oil and gas operations, as well as provide a cleaner, more environmentally friendly produced water that is discharged into the sea.
Conclusion
In Brazil, due to changes in the required regulatory practices for detecting TOG in produced water, the water-soluble naphthenic acids are now included in the TOG measurement for oceanic overboard discharge. The current TOG measurement process includes hexane extraction that occurs at a pH of 2. In the process of lowering the pH, a portion of the NAs is driven from the water phase into the hexane organic phase during analysis. In the typical produced water treatment process the pH remains higher which contributes to more NA being discharged overboard.
Acid pH manipulation of the produced water stream is one possible solution for improved naphthenic acid removal, but it is environmentally and economically prohibitive. In order for energy and services companies to meet the demands of the new Brazilian regulatory regime, and in order to prepare for potential changes in regulatory regimes in other countries with similar oil profiles, it will be necessary for these companies to explore new and innovative water treatment technologies that can offer both the capture and removal of NA with the least need for pH adjustment.
Attention will be needed to determine where in the complete process train these new technologies should exist, as the complete treatment train should be considered. With a new method for analyzing overboard discharge comes the need for a new way of thinking about produced water treatment, and the need for innovative companies and technologies to provide those solutions. WT
References
1. Bertheussen, Are. “Naphthenic Acid Solubility in Produced Water and Their Interactions with Divalent Cations,” Journal Norwegian University of Science and Technology (NTNU), 2018, series 259. https://ntnuopen.ntnu.no/ntnu-xmlui/ handle/11250/2565557
2. Bertheussen, Are; Simon, S.; Sjoblom, J. “Equilibrium Partitioning of Naphthenic Acid Mixture Part 2: Crude Oil-Extracted Naphthenic Acids,” Journal Energy Fuels, 2018, pp 9142-9158. https://pubs.acs.org/doi/10.1021/acs.energyfuels.8b01494
About the Author
Caleb Smathers
Caleb Smathers is the subject matter expert (SME) on ProSep’s Osorb Media System (OMS) and leads other key product development efforts. He has co-authored several patents/patent applications, peer-reviewed and other publications, and has presented at prominent conferences on produced water treatment.
John Sabey
John Sabey, one of the original cofounders of ProSep, is responsible for providing leadership and mentorship to ProSep’s technical team. With more than 30 years of experience in engineering, sales, technology deployment, and product development in the oil and gas sector, his principal focus is driving the adoption of high-efficiency mixers to the midstream, natural gas processing, downstream and LNG segments.
Amanda Reichert
Amanda Reichert has six years of experience in the oil and gas industry working as a process engineer for engineering and consulting companies in both Brazil and the U.S. She joined ProSep in 2019 as a process engineer and has been working in the development of proprietary technologies.
Gregory Hallahan
Gregory Hallahan has more than 13 years of experience in the oil and gas industry. In his previous role as director of product development, he led the push of ProSep’s proprietary technologies out into the marketplace. Hallahan currently oversees the operations of the company as the chief operating officer.